Next Lesson - Urinary Tract Obstruction
Abstract
- Chronic Kidney Disease (CKD) is a long-term reduction in renal function over a period of months to years, which is often progressive and may be irreversible. CKD can eventually progress to end-stage renal failure with treatment aiming to slow the progression of the disease.
- CKD is diagnosed using the glomerular filtration rate (GFR) and is defined as a reduced GFR for 3 months or more. It is staged using the GFR and the level of proteinuria.
- Common histological changes seen in CKD include a fibrotic cortex, sclerotic glomeruli, scattered inflammatory cells and thickened arteries.
- Some of the causes of CKD include diabetes, hypertension, glomerulonephritis, chronic or recurrent tubular injury, idiopathic causes and chronic infection.
- CKD investigations aim to define the degree of renal impairment, identify the cause and assess for complications of chronic kidney disease.
- The main aim of CKD management is to prevent the progression of the disease and to treat any complications that develop. Management includes hypertension control, reducing the level of proteinuria and controlling diabetes.
- Chronic kidney disease has many complications and these must be looked out for because they can increase the mortality and morbidity of patients. Some complications include hyperkalaemia, acidosis, anaemia and mineral bone disease.
- End-stage renal disease is the final stage of CKD where death is likely without renal replacement therapy. It is defined as a GFR < 15 ml/min.
- End-stage renal disease is treated with renal replacement therapy and kidney transplant. If a patient does not choose either of these two treatments then they are managed with active conservative therapy.
- Dialysis is the main renal replacement therapy used. In dialysis waste products and excess fluid are removed from the body when the kidneys stop functioning.
- Active conservative management is where patients are managed by a multidisciplinary team approach with the main management being symptom control, advanced care plans and supportive care for patients and their family.
- Haemodialysis involves pumping blood out of the body and into a dialyser to filter the blood to remove excess fluid and waste products. Patients on haemodialysis have lots of restrictions on their fluid intake and what they can eat.
- Peritoneal dialysis involves using the peritoneal cavity to exchange substances between the dialysate and the blood. The peritoneal cavity is filled with dialysate and left for a while to allow for the exchange of substances.
- Kidney transplantation is for patients who have little or no kidney function where the patient receives a healthy kidney from another individual. The kidney can come from a living related donor, a living unrelated donor or a deceased donor.
Core
Chronic kidney disease (CKD) refers to abnormalities of renal structure or function present for 3 months or more, which may be progressive. It is more common in the elderly, ethnic minorities, those with multiple co-morbidities and those who are socially deprived. Patients with CKD have an increased risk of developing acute kidney injury because the kidneys are already damaged and so can be damaged more easily.
It is diagnosed using the glomerular filtration rate (GFR), with a normal GFR being 90-120 ml/min/1.73m2. In CKD there is a lowered GFR because not enough fluid is being filtered by the kidneys due to the loss of renal function.
CKD is defined as a reduced GFR that persists for 3 months or more.
In CKD, damaged renal tissue is replaced by an extracellular matrix, resulting in decreased amount of functioning kidney tissue. The disease will eventually progress to scarring of the kidneys at the end of CKD. Histologically, there are four main features seen on a biopsy: a fibrotic cortex, sclerotic glomeruli, scattered inflammatory cells and thickened arteries.
Usually patients with CKD are asymptomatic, so if there are any risk factors present, the patient will be investigated with blood tests with the aim to treat and manage the patient to prevent renal failure from developing. Any potential symptoms are usually non-specific and can include fatigue, nausea, pruritus (itching) and nocturia.
Chronic kidney disease may progress over time to end-stage renal failure, and the main aim of treatment is to slow disease progression and reduce complications.
Stages of Chronic Kidney Disease
Chronic kidney disease is staged using the GFR and the quantity of proteinuria. The lower the GFR and the more severe the proteinuria, the worse the prognosis. The following table shows the stages of CKD along with the GFR and the proteinuria levels for each stage.
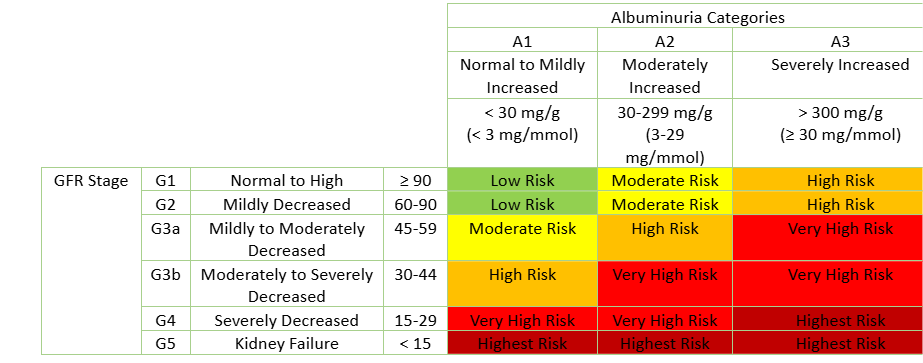
Table - The Stages of CKD
SimpleMed original by Dr. Thomas Burnell
Causes of Chronic Kidney Disease
CKD arises as a result of another condition damaging the kidneys over time and so there are many causes because a lot of conditions can affect the kidneys. Examples of these conditions include:
- Diabetes
- Hypertension
- Glomerulonephritis
- Infection - e.g. pyelonephritis
- Genetic - e.g. autosomal dominant polycystic kidney disease, Alport’s syndrome
- Obstruction and reflux nephropathy
- Acute tubular injury
- Vascular disease - e.g. vasculitis
- Idiopathic - CKD may just develop over time in an otherwise healthy patient with no known cause.
When investigating chronic kidney disease, it is important to identify the degree of renal impairment, the cause of renal impairment and to look for the complications of CKD.
The degree of renal impairment is identified using:
- Blood pressure
- Urinalysis
- Creatinine and eGFR - these will help to define the stage of CKD.
- Other blood tests: urea and electrolytes, full blood count, CRP level, liver function tests, iron levels, bone biochemistry
The cause of renal impairment is identified using different techniques for different differential diagnosis:
- Autoimmune disease - an autoantibody screen and complement levels can be used to look for autoimmune disease.
- Vasculitis - anti-neutrophil cytoplasmic antibody screen can be used to look for vasculitis.
- Myeloma - myeloma can cause CKD and the main investigations into myeloma are serum immunoglobulin screen, protein electrophoresis and serum free light chain levels.
- Ultrasound scan - ultrasound scans can be used to see the kidney size and look for evidence of obstruction. Kidneys are typically smaller in CKD.
- Kidney biopsy - this can be used if the cause of chronic kidney disease remains unknown after other investigations.
- Stones and masses - these can be investigated for using a CT scan or an MRI scan.
- Renal artery stenosis - an MR angiogram can be used to look for a renal artery stenosis.
Identifying complications of CKD:
- Full blood count - this can be used to look for anaemia.
- Assessment of mineral and bone disorder (primarily with blood tests)
- Bone biochemistry - there will be a decreased serum calcium, an increased serum phosphate and increased parathyroid hormone.
The main aim in management of these patients is to prevent the development of CKD in the first place. Once a patient develops CKD, the aim of treatment is to prevent the progression of the disease and to treat any complications that develop. The main factors in the prevention and treatment of CKD is through addressing modifiable risk factors, controlling hypertension and controlling the level of proteinuria.
- Modifiable Risk Factors:
- Smoking - advise patients to stop smoking.
- Obesity - advise patients to lose weight and do more exercise.
- Hypertension Control:
- Anti-hypertensive medication - ACE inhibitors and angiotensin receptor blockers are typically used because of their anti-proteinuria effect.
- Diuretics
- Fluid restriction
- Proteinuria - This should be kept as low as possible in patients with CKD to prevent progression of the disease.
- ACE inhibitors and angiotensin receptor blockers are the main medications used to treat proteinuria as they reduce the glomerular filtration pressure and so decrease the amount of protein being filtered.
- Diabetes - patients with diabetes should have well controlled HbA1c to prevent CKD progressing.
- Lipid levels - there tends to be a high cholesterol level in CKD so patients should be treated with lipid lowering medication (statins) to reduce the risks of high cholesterol.
There are many complications of chronic kidney disease and these must all be identified and treated to reduce the risk of further complications and improve patient’s quality of life. Some of the complications include hyperkalaemia, acidosis, anaemia, mineral bone disease and altered drug metabolism.
In chronic kidney disease there is reduced excretion of potassiumAs less potassium is being removed from the body the levels in the blood riseleading to hyperkalaemia. To reduce the risk of hyperkalaemia, potassium intake and contributing medications are reviewed, and management is guided by renal function and overall fluid status.
If a patient’s serum potassium level is increasing, it may require the avoidance of drugs that increase potassium levels and a diet alteration to avoid foods high in potassium.
- ACE inhibitors and angiotensin receptor blockers used in the treatment of CKD may need dose adjustment or closer monitoring if serum potassium rises, and are stopped if hyperkalaemia is significant or persistent.
In chronic kidney disease the reduced kidney function leads to a decreased excretion of acid from the body and an increased loss of bicarbonate. This leads to a metabolic acidosis as the control of blood pH is lost. This acidosis will happen slowly and may not be noticed because the symptoms are quite non-specific.
Symptoms of Acidosis include:
- Headache
- Drowsiness
- Diarrhoea
- Muscle weakness
- Coughing
- Nausea and vomiting
- Shortness of breath
- Confusion
- Seizures
Acidosis must be treated because it can lead to worsening renal function and muscle breakdown. It can cause a loss of bone minerality, which can further exacerbate mineral bone disease in CKD.
The main treatment of acidosis is oral sodium bicarbonate (NaHCO3), which will replace the bicarbonate that is being lost from the body. Bicarbonate supplements will help to prevent the pH from decreasing, and becoming acidotic, by neutralising the excess acid in the blood.
Anaemia is caused by multiple mechanisms in chronic kidney disease, all of which act to lower haemoglobin and cause anaemia. Some of the mechanisms that cause anaemia in CKD are:
- Acidosis - a low pH decreases the production of erythropoietin leading to reduced red blood cells production and anaemia.
- Anaemia of chronic disease - inflammation causes a high hepcidin level in the body which decreases free iron levels in the body causing anaemia.
- Shorter red blood cell lifespan
- Medication
- CKD mineral and bone disease
- Deficiency of Vitamin B12, folate, iron
The treatment of anaemia is very important as it improves the patient’s quality of life and reduces patient mortality. Treating anaemia helps to improve exercise capacity and cognitive function as well as slowing the progression of renal disease. It can also prevent left ventricular hypertrophy, as the heart has to pump harder to deliver enough oxygen around the body to counter the effects of anaemia, leading to strain on the heart, especially the left ventricle.
Before treating anaemia iron stores (ferritin) and serum iron levels should be checked, as a low iron level may be the cause of anaemia. Folate and Vitamin B12 should also be checked to look for deficiencies. If any of these are found to be low then the appropriate treatment should be taken to increase the levels back to normal, e.g. oral folate supplements, Vitamin B12 injections.
If iron, folate and Vitamin B12 are all found to be fine but the patient is still anaemic, then an erythropoietin stimulating agent can be given to increase haemoglobin levels in the body. The aim of this treatment is not to get a normal or perfect level but to achieve a level of haemoglobin that does not cause the patient any symptoms.
Mineral bone disease is seen in chronic kidney disease as the kidneys become unable to regulate calcium and phosphate levels within the body. There are many causes of mineral bone disease which all combine to cause a decreased calcium level and reduced bone mineralisation within the body.
Some of the causes of mineral bone disease include:
- Impaired renal function which decreases phosphate excretion from the body, resulting in an increased serum phosphate (hyperphosphataemia). Hyperphosphataemia stimulates the parathyroid glands leading to an increase in parathyroid hormone secretion and increased bone reabsorption, reducing bone breakdown and decreasing serum calcium levels.
- Decreased levels of activated Vitamin D due to a reduced ability of the kidneys to hydroxylate 25-hydroxyvitamin D. This reduction in Vitamin D levels impairs bone mineralisation and calcium absorption.
- Increased phosphate levels within the body causes phosphate in the blood to combine with free calcium resulting in calcium deposits precipitating throughout the body. This decreases the concentration of free calcium within the body and so reduces bone mineralisation and building to counter the reduced calcium level.
Mineral bone disease in CKD can manifest itself as osteomalacia, osteosclerosis or osteoporosis. The blood results of a patient with mineral bone disease will be a secondary hyperparathyroidism picture:
- Low serum Vitamin D
- Low serum Calcium
- High serum Phosphate
- High serum Alkaline phosphatase
- High serum Parathyroid Hormone
Mineral bone disease is managed through reduced phosphate intake, the use of phosphate binders and synthetic Vitamin D injections (1-α-calcidol). These methods help to increase serum calcium levels within the body and to reduce the phosphate levels to prevent the precipitation of calcium deposits and other complications of hyperphosphataemia.
The kidneys are one of the main places in the body where drug excretion and metabolism occur, hence in CKD there can be reduced excretion of drugs and their metabolites resulting in increased risk of toxicity due to their accumulation.
Because of reduced drug excretion in CKD, many drugs require dose reduction to prevent their levels getting too high and causing toxicity. This is especially important in nephrotoxic drugs as these can further damage the kidney and worsen CKD.
Other Complications - some other complications that occur with CKD include uraemic syndrome, hyperparathyroidism, fluid overload, electrolyte disturbances, malnutrition and cardiovascular disease.
- Waste products can accumulate in the body due to reduced excretion by the kidney which contribute to uraemic syndrome, reduced appetite, nausea and vomiting, and pruritus.
End Stage Renal Disease/Failure
End-stage renal failure is the final stage of chronic kidney disease and is a result of extensive damage to the kidneys. Death is likely without renal replacement therapy. It is defined as an estimated GFR < 15 ml/min/1.73 m2, persisting chronically.
Non-specific symptoms of end-stage renal failure may not be recognised, so it is important to check the GFR of patients with CKD. Symptoms of CKD include:
- Fatigue
- Symptoms of volume overload, e.g. peripheral oedema (swelling of the ankles)
- Nausea and vomiting
- Reduced appetite
- Restless legs and cramps
- Pruritus (itching)
- Sexual dysfunction and reduced fertility
- Increased risk of infections due to reduced cellular and humoral immunity
Treatment of end-stage renal failure is through renal replacement therapy and kidney transplant. Dialysis is the main renal replacement therapy used to treat end-stage renal failure. Patients may also choose active conservative therapy if they decide not to have renal replacement therapy or kidney transplant. The possible treatments of end-stage renal failure should be discussed with the patient before they reach renal failure so that they understand the advantages and disadvantages of each option before they are required.
Dialysis is the method by which waste products and excess fluid are removed from the body when the kidneys stop functioning. It involves the use of a dialysis machine to filter the blood either by diverting the blood out of the body to be filtered (haemodialysis) or using a membrane in the body to remove substances and fluid from the blood (peritoneal dialysis).
- There are two main types of dialysis, haemodialysis (HD) and peritoneal dialysis (PD) which are discussed below.
- There are three other types of dialysis which are haemofiltration, haemodiafiltration and intestinal dialysis, but they will not be discussed in this article
Active Conservative Management
This treatment option for end-stage renal failure is used for patients who are either unsuitable for or choose not to have invasive therapy such as peritoneal dialysis, haemodialysis or kidney transplant. This complex decision is usually made after multiple discussions with the patient and their family.
In this option, patients are managed under a multidisciplinary team approach via symptom control, advanced care plans, supportive care for patients and their family, and ensuring the patient remains in their preferred place of care.
Haemodialysis involves pumping blood out of the body and into a machine, called a dialyser, that filters the blood to remove excess fluid and waste products. It is carried out in the hospital three times a week at designated slots, and lasts around 4 hours. The process requires the patient to take a lot of medication and to have an arteriovenous fistula (connection between an artery and a vein) put into their arm where the dialysis machine can be attached to.
Patients also have large restrictions on their diet as they are restricted to a small volume of fluid per day and must avoid foods high in potassium and salt.
Haemodialysis can also be performed at home which allows for a little more freedom and less disturbance to daily life. There are three types of home haemodialysis and patients can choose which type they would prefer to do. Usually patients on home haemodialysis are able to undergo dialysis for longer periods of time allowing better molecule clearance which improves their symptoms. However, home haemodialysis does require training and the ability to use the machines, as well as someone being at home to make sure it functions correctly.
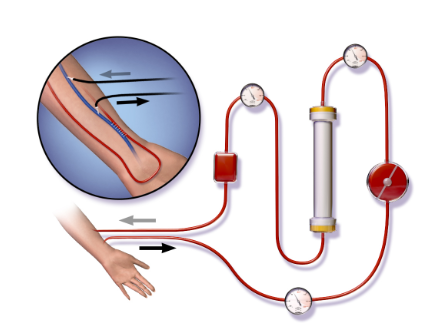
Diagram - The Process of Haemodialysis
Licensed under Creative Commons, Source.
- Failed vascular access - if the patient cannot have a fistula put in.
- Heart failure - haemodialysis can cause rapid fluid and haemodynamic shifts which may worsen cardiovascular instability. However, heart failure is not an absolute contraindication, and haemodialysis is commonly used with careful monitoring and adjustment of fluid removal.
- Coagulopathy
- Infection
- Thrombosis
- Venous stenosis
- Haemorrhage
- Steal syndrome
- Anaemia
- Cardiovascular instability
- Cardiac arrhythmias
- Air embolism
- Nausea and vomiting
Peritoneal dialysis involves using the peritoneal cavity to exchange substances between dialysate and the blood. Dialysate is the fluid used in peritoneal dialysis to allow waste products to diffuse from the blood, while ultrafiltration removes excess fluid.contains dextrose (a glucose solution) which draws fluid and waste from the blood, and keeps it in the solution. Once dialysis is complete, the dialysate is disposed of and a new bag is used the next time.
There are two main types, continuous ambulatory peritoneal dialysis (CAPD) and automated peritoneal dialysis (APD), and both of which involve pumping dialysate into the peritoneal cavity where substances can then be exchanged between the fluid and the blood.
Continuous ambulatory peritoneal dialysis involves four sessions of peritoneal dialysis per day where the patient’s abdominal cavity is filled with a bag of dialysate over 40 minutes. The solution is then left in the abdomen for a few hours before being drained out. The process requires the patient to have a catheter inserted into their abdomen to fill and drain the abdominal cavity of dialysate. Once the used fluid is drained, the abdomen is filled with new fluid.
CAPD can be carried out anywhere, either at home or work, and requires the patient to be trained in how to perform CAPD. Exchanges can happen at any time of the day but most patients choose to do them before meals and before bed.
Automated peritoneal dialysis involves one session of peritoneal dialysis per day where the patient is attached to a machine for around 9 hours. This is usually carried out at home and requires the patient to be trained in how to use the machine. Most people choose to do their dialysis overnight due to the length of time required for APD.
When compared to haemodialysis there are fewer fluid and diet restrictions, yet dietitians are involved in deciding any restrictions that the patient must have.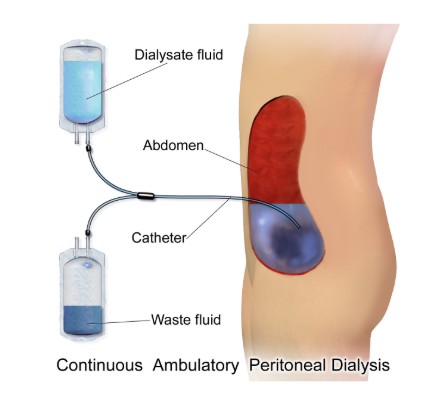
Diagram - The Process of Continuous Ambulatory Peritoneal Dialysis, with old dialysate fluid being exchanged for new fluid
Licensed under Creative Commons, Source.
- Failure of peritoneal membrane - peritoneal dialysis causes scarring of the peritoneal membrane which reduces diffusion of waste across the membrane.
- Intra-abdominal adhesions - these will prevent the peritoneal dialysis from working.
- Previous abdominal surgery, hernia or stoma - these may be associated with peritoneal scarring and can make peritoneal dialysis more difficult, but suitability depends on individual assessment.
- Obesity or large muscle mass - peritoneal dialysis requires a large peritoneum surface area to body volume ratio.
- Patient or carer unable to connect or disconnect the peritoneal dialysis
- Peritonitis
- Catheter associated problems - e.g. infection, blockage, kinking and leaks.
- Constipation
- Fluid retention
- Hyperglycaemia
- Weight gain
- Ultrafiltration failure
- Hernia
- Scarring of the peritoneum
A kidney transplant is where a patient who has little or no kidney function (e.g. end-stage renal failure) receives a healthy kidney from another individual. This kidney can either come from a live or a deceased donorbut they must be blood group compatible, and closer tissue matching improves transplant outcomes.replacement kidney is inserted into the lower abdominal cavity and then the renal vessels and ureter are surgically attached to it to allow the kidney to function as normal.
Patients who undergo a renal transplant have both a greater life expectancy and quality of life compared to patients on dialysis. Transplantation means the patient can stop dialysis, and it also can help to reverse anaemia and renal bone disease.
Contraindications for Transplantation
- Active infection or malignancy
- Active viral hepatitis
- Severe heart or lung disease
- Reversible kidney disease
- Uncontrolled substance abuse
- Psychiatric illness
- Short life expectancy
- Non-adherence to ongoing treatment
- Complications during operation - e.g. local infection and deep vein thrombosis.
- Immediate graft failure - where the transplanted kidney does not function from the outset, for example due to vascular thrombosis or primary non-function; hyperacute rejection is a rare cause.
- Infection
- Cancer
- Side-effects from immunosuppressive drugs
Renal transplants require lots of medication after the operation to prevent rejection of the transplanted kidney, and many of these are immunosuppressants as these suppress the immune system to prevent it from attacking the new kidney. Some side effects from these medications include:
- Hypertension
- Gastrointestinal tract ulceration
- Nausea
- Diarrhoea
- Thrombocytopenia
- Leukopenia
- Diabetes mellitus
- Hypercholesterolaemia
The kidneys that are used in transplant come from three main donor sources:
- Living Related Donor - this is the best possible source of a kidney as related donors have a better compatibility than unrelated donors, so there is a reduced risk of rejection by the body.
- Living Unrelated Donor - with unrelated donor kidneys, there is a similar outcome to related donor kidneys, but they are still not as good as related donors.
- Deceased Donor - kidneys from decreased donors have the lowest survival compared to living related and unrelated donors. There is also a smaller window for the kidneys to be transplanted as the procedure must be done quickly as the kidneys will not survive long outside of the body.
Patients who have had a kidney transplant require long term follow-up care several times a month for the first few months to ensure nothing has gone wrong. They then require follow-up appointments every so often to check the function of the kidney. They are also screened for malignancies, offered the influenza and pneumococcal vaccine, and have regular blood tests (e.g. calcium, phosphate and glucose levels) to monitor for complications (e.g. mineral bone disease and diabetes).
Complications of Transplant and Immunosuppressive Treatment
- Post-operative complications - e.g. bleeding, deep vein thrombosis, pulmonary embolism, local infection and pain.
- Infections - especially opportunistic infections that can occur as a result of immunosuppressive treatment.
- Cancer
- Drug toxicity
- Bone marrow suppression - this can occur as a result of the immunosuppressive treatment.
- Urinary tract obstruction
- Graft rejection - graft rejection can be hyperacute, accelerated, acute cellular or chronic. Most of these can be treated with immunosuppressive treatment.
Edited by: Dr. Tom Bradley
Reviewed by: Adrian Judge
- 516

