Next Lesson - Lung Function Testing
Abstract
- Hypercapnia occurs when there is a rise in the partial pressure of carbon dioxide above 6kPa, hypocapnia occurs when there is a reduction in the partial pressure of carbon dioxide below 4.7kPa.
- Haemoglobin oxygen saturation decreases when pO2 falls below 8kPa.
- When pCO2 increases, the pH of the plasma will fall, causing acidosis. When pCO2 decreases, the pH of the plasma will rise, causing alkalosis.
- The kidneys can excrete less or more hydrogen carbonate ions depending on the concentration of hydrogen ions present in the plasma of the blood.
- The body produces acids in various metabolic processes, which consume hydrogen carbonate ions during buffering reactions and can lower plasma pH. This lowers the plasma pH and deemed as metabolic acidosis.
- When a patient persistently vomits, there is a large loss of hydrogen ions from the stomach acid, causing the plasma pH to increase. This is deemed as metabolic alkalosis.
- There are chemoreceptors located in the aortic arch and carotid bodies that are sensitive to changes in pO2.
- The central chemoreceptors are located in the medulla and are exposed to the cerebrospinal fluid. The chemoreceptors respond to changes in CSF pH (controlled by pCO2) and the blood-brain barrier separates the CSF from the blood.
- Serum potassium concentrations can alter depending on the pH of the plasma.
Core
The pH of plasma is important because having a high or low pH can affect the ability of certain proteins to function. For example, haemoglobin molecules function best at pH around 7.4, meaning that the pH of plasma needs to be kept between 7.35 and 7.45 to allow adequate oxygen transport.
In clinical practice, this is measured on an arterial blood gas (ABG). The interpretation of this test is explained at the end of the article (it is important to understand the physiology behind the control of plasma pH to be able to interpret an ABG).
Hypercapnia occurs when there is a rise in the partial pressure of carbon dioxide (pCO2) above 6kPa, whilst hypocapnia occurs when there is a reduction in the partial pressure of carbon dioxide below 4.7kPa.
Hypoxia is when there is a lower than normal of oxygen in the arterial blood, meaning there is a reduction in the amount of oxygen reaching metabolic tissues. The normal reference range for the partial pressure of oxygen (pO2) on an arterial blood gas is 11-13kPa.
When there is an increased ventilation rate, the pO2 will increase and the pCO2 will fall as more oxygen is inhaled and more carbon dioxide is expired. The opposite can be seen when the ventilation rate decreases.
In times of hypoxia, the ventilation rate will increase (hyperventilation) as an attempt to increase the pO2, but this leads to a decrease in the pCO2.
Based on the oxygen-haemoglobin disassociation curve, the oxygen saturation of haemoglobin falls much more steeply below 8kPa. This means that at any partial pressure of oxygen above 8kPa, haemoglobin is pretty much fully saturated. However, due to the co-operative binding seen with haemoglobin (change in structure of haemoglobin as more oxygen molecules bind to make it easier to fully saturate), below 8kPa there is a steep drop in percentage saturation of haemoglobin.
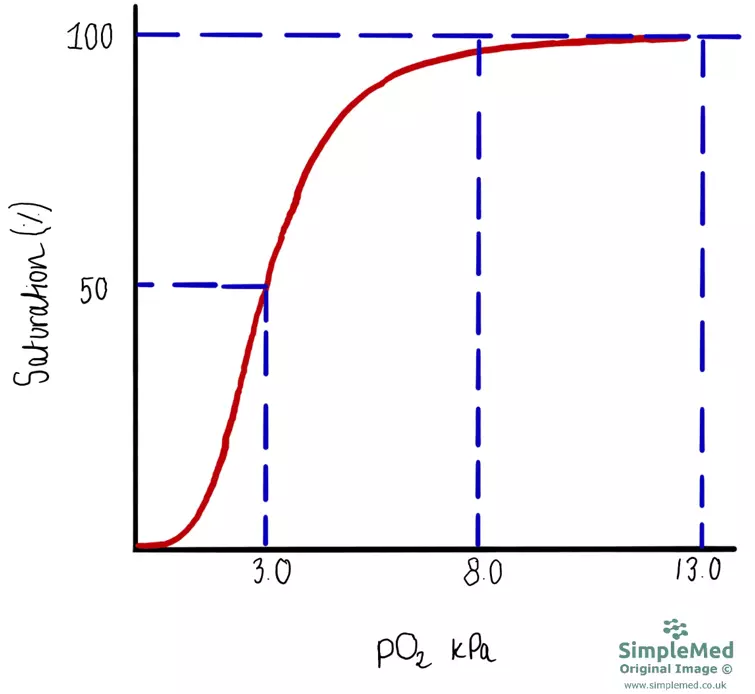
Diagram - Graph of the oxygen-haemoglobin disassociation curve, showing that saturation is only affected below 8kPa
SimpleMed original by Dr. Peter Parkinson
Once the pCO2 begins to increase, the pH of the plasma will begin to fall, causing acidosis. When the pCO2 begins to decrease, the pH of the plasma will increase, resulting in alkalosis.
Structures and mechanisms within the body can be affected by changes to pH of the plasma. Enzymes can lose normal function and may denature at extremes of pH, which can affect processes that rely on enzymes such as aerobic respiration and gluconeogenesis.
Once the pH of the plasma is above 7.6, the distribution of free calcium ions changes, with less ionised calcium available due to increased binding to proteins. This increases neuronal excitability, making neurones more likely to depolarise and can cause tetany (involuntary muscle contraction).
Hypoventilation can cause a build up of carbon dioxide in the blood. When this dissociates, it produces hydrogen (H+) ions, reducing the pH of the blood. This is known as respiratory acidosis. Conditions such as type 2 respiratory failure, e.g. severe chronic obstructive pulmonary disease (COPD), drug overdose and neuromuscular disease, can lead to respiratory acidosis.
Hyperventilation causes the opposite, because all the carbon dioxide is breathed out. This causes H+ and hydrogen carbonate (HCO3-) ions to come together to replace the lost CO2, which removes H+ ions from the blood, causing a respiratory alkalosis. Conditions such as panic attacks can cause respiratory alkalosis.
There are two partial pressure being controlled during the process of breathing: oxygen and carbon dioxide. There is less precise control of the partial pressure of oxygen as long as it stays above around 8 kPa, which is usually sufficient to maintain high oxygen saturation. There are peripheral chemoreceptors located in the aortic arch and carotid bodies that are sensitive to a decline in pO2. By having a high rate of blood flow, these structures only respond to low pO2 and once stimulated, they cause a number of effects: increased respiratory rate, increased blood flow to the brain and kidneys, and increased heart rate.
The peripheral chemoreceptors that are sensitive to changes in the partial pressure of carbon dioxide, are located in aortic arch and carotid bodies. They are less sensitive compared to central chemoreceptors, as they need a big increase of carbon dioxide (1.3kPa) to do anything.
The central chemoreceptors are located in the medulla of the brainstem and are exposed to the cerebrospinal fluid (CSF). These chemoreceptors respond to changes in CSF pH. Carbon dioxide is able to diffuse into the CSF from the blood, but hydrogen carbonate and hydrogen ions are unable to. This means that the central chemoreceptors are exposed to the concentration of dissolved carbon dioxide only, not the concentrations of other ions.
The choroid plexus is a specialised structure that produces and helps regulate the composition of CSF, including the movement of HCO3- ions into and out of the CSF. The blood brain barrier itself is formed by specialised capillary endothelial cells. These cells are found in the ventricles of the brain and communicate with the CSF.
This system is independent of the concentration of hydrogen carbonate ions in the blood. The pH of the CSF (like the plasma) is determined by the ratio between concentration of hydrogen carbonate ions to concentration of dissolved carbon dioxide. Any rapid changes in the concentration of plasma carbon dioxide will take time to alter the concentration of carbon dioxide in CSF, because diffusion across the blood brain barrier takes time.
If arterial pCO2 rises, there will be a short delay before the CSF pCO2 increases. This change will be detected by central chemoreceptors and will lead to an increase in ventilation to attempt to remove the excess carbon dioxide. This is a form of negative feedback.
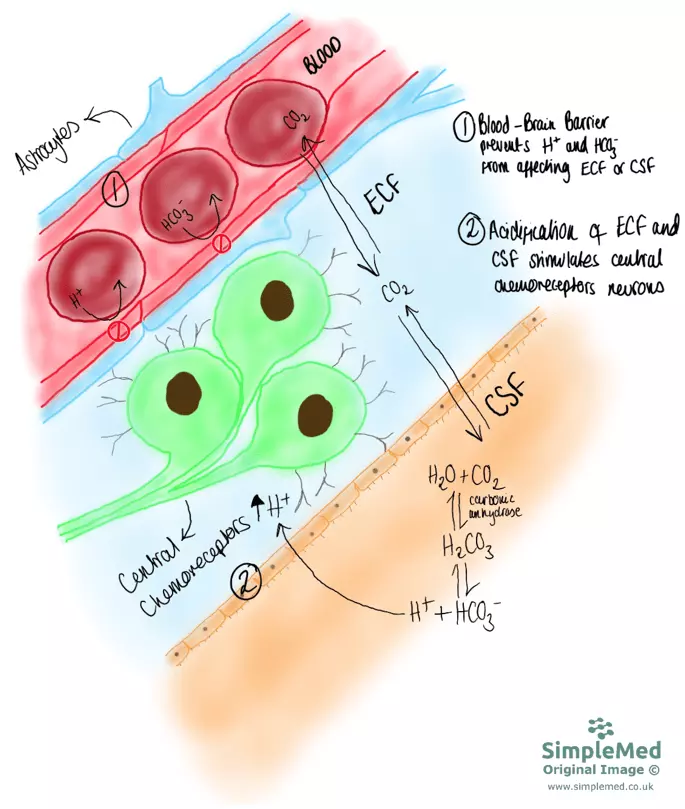
Diagram - Central chemoreceptors and how they react to the increased hydrogen ions in cerebrospinal fluid and extracellular fluid, second to an increased plasma pCO2
SimpleMed original by Dr. Peter Parkinson
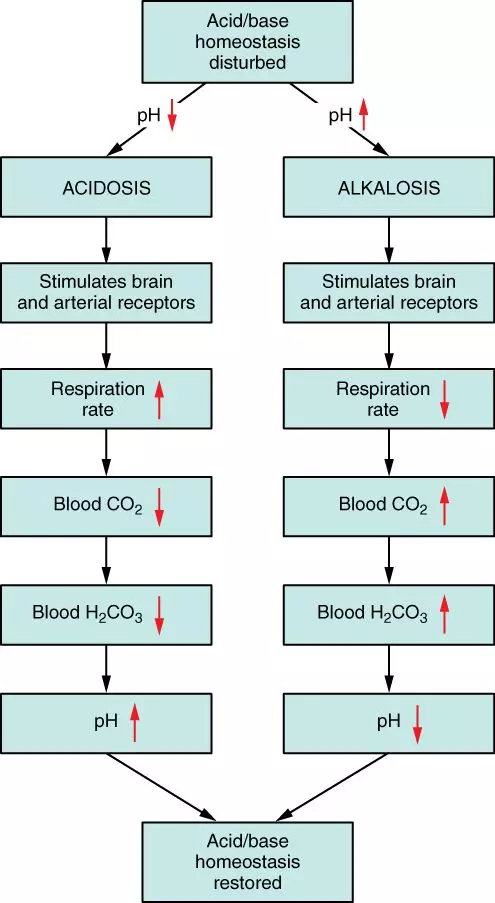
Diagram - The homeostatic control of plasma pH and the responses made to changes to pH
Creative commons source by OpenStax College [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)]
In people with healthy lungs, this system works really well as changes in pCO2 are detected centrally and the ventilation rate changed accordingly.
However, in people with chronic lung conditions, like COPD, increased levels of CO2 become normal as their lungs are not very good at getting rid of CO2. If the system worked in these patients as it does in a healthy patient, they would be persistently hyperventilating, which would be very uncomfortable.
In reality, with cases of chronic hypercapnia, the CSF pH prompts the choroid plexus to persistently pump in large amounts of HCO3- ions, meaning that the pH becomes normal. This brings the ratio of carbon dioxide to hydrogen carbonate into normal range (even though both of these results are high), and this means that the pH is normal. This returns the breathing rate of this patient to normal, and ‘resets’ the system around a higher tolerated pCO2.
This ‘resetting’ of the central chemoreceptors to accept much higher pCO2 as ‘normal’ means that the patient depends on their oxygen sensitive peripheral chemoreceptors to modify their ventilation rate if needed. For example, in a patient with COPD, their central chemoreceptors may have ‘reset’, but their peripheral chemoreceptors still detect a low pO2, so the ventilation rate is increased. This is known as the ‘hypoxic drive’, and is why, in some COPD patients, giving high levels of oxygen for long periods of time can be dangerous. Raising the pO2 reduces peripheral chemoreceptor stimulation, which can contribute to reduced ventilation.
Metabolic Acidosis and Alkalosis
The body produces acids in various processes: lactic acid during anaerobic respiration and acidic ketones from gluconeogenesis of fatty acids and ketogenic amino acids.
Hydrogen carbonate ions react with the H+ ions produced by the body and when in excess, it can lead to the depletion of HCO3- ions. As the concentration of H+ ions increases, the ratio between pCO2 and HCO3- ions increases, resulting in a decrease in plasma pH. This is deemed as metabolic acidosis. The body tries to counteract the decrease in plasma pH by increasing ventilation to decrease pCO2 and bring the plasma pH into the normal range.
When a patient persistently vomits, there is a large loss of H+ ions from the body, causing the plasma pH to increase. This is deemed as metabolic alkalosis. The body attempts to compensate by decreasing ventilation to reduce the volume of carbon dioxide expired, to try to bring the plasma pH in the normal range. However, the lungs also need to maintain O2 levels, meaning that this is rarely possible. This means that the kidneys are mostly responsible for compensation of the alkalosis, by increasing the excretion of HCO3- ions in the urine. This is very effective but is not a fast compensatory mechanism.
Renal Production and Control of Hydrogen Carbonate Ions
The pH of the plasma depends on the ratio of the concentration of HCO3- ions to the pCO2 of the blood. The kidneys are responsible for controlling the concentration of HCO3- ions by altering the excretion of the ion.
Hydrogen carbonate ions are filtered at the glomerulus of the kidneys and enter the filtrate, but the majority of these ions are recovered at the proximal convoluted tubule (PCT). PCT cells excrete H+ ions into the lumen as they transport sodium ions into the cell. The H+ ions react with HCO3- ions present in the filtrate, resulting in carbon dioxide and water being formed. The carbon dioxide diffuses into the PCT cells and the reverse reaction occurs, producing H+ and HCO3- ions inside the PCT cell. The HCO3- ions can then be secreted into the extracellular fluid (ECF) before moving into the plasma.
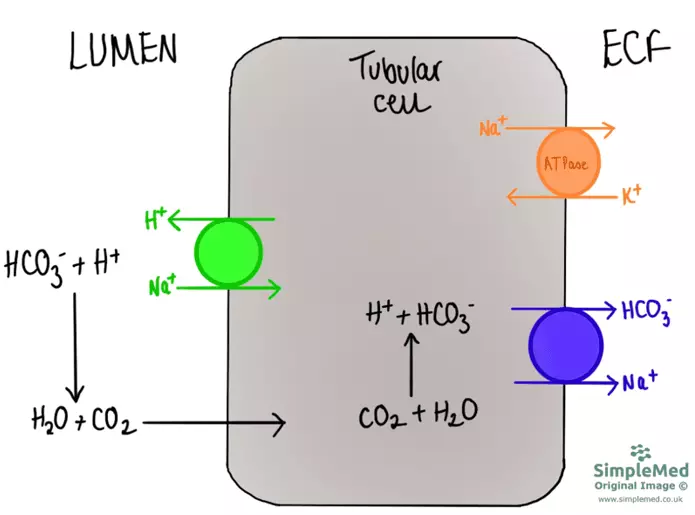
Diagram - The movement of hydrogen ions out of proximal convoluted tubule (PCT) cells and movement of hydrogen carbonate ions into the extracellular fluid from PCT cells
SimpleMed original by Dr. Peter Parkinson
Kidneys also produce HCO3- ions from amino acids, in the proximal tubule cells of the kidney, but this process takes a while. Glutamine (an amino acid) is broken down to ammonium (which further breaks down into H+ ions and ammonia) and α-ketoglutarate. The ammonia molecules diffuse into the filtrate and are able to bind with H+ ions present in the lumen to form ammonium, which cannot diffuse across the cell membrane, trapping the ammonium in the filtrate. This is a form of buffering in the urine to prevent the urine from becoming too acidic, as this would damage the cells of the urinary tract. The α-ketoglutarate is converted to HCO3- ions inside the cells of the PCT, which can be secreted into the ECF, before being transported into the blood.
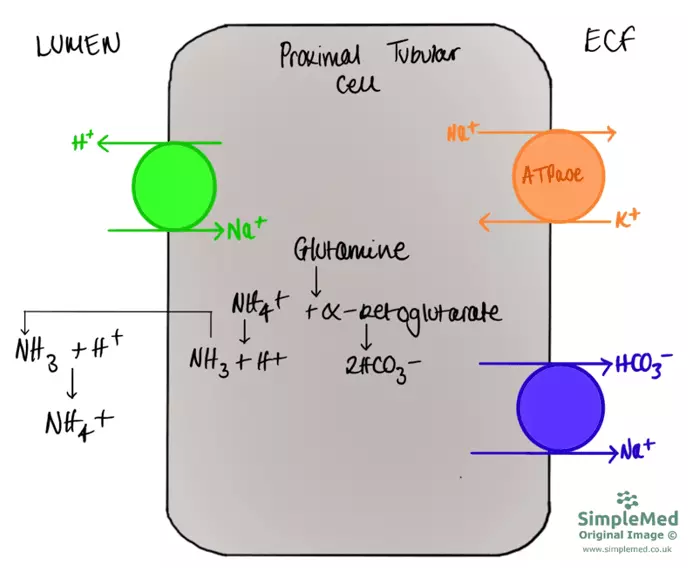
Diagram - The generation of ammonium and hydrogen carbonate ions from glutamine
SimpleMed original by Dr. Peter Parkinson
In the distal convoluted tubule and collecting ducts, H+ ions are also actively secreted into the lumen via α-intercalated cells. These H+ ions bind with ammonia and phosphate ions; these molecules are then excreted in urine. Phosphate ions act in the same as as ammonia, by buffering H+ ions.
Inside the α-intercalated cells, the reaction of carbon dioxide and water to H+ ions and HCO3- ions is in favour of the forward reaction as H+ ions are excreted into the lumen. Hydrogen carbonate ions are secreted into the ECF by α-intercalated cells as chloride ions are moved into the α-intercalated cells.
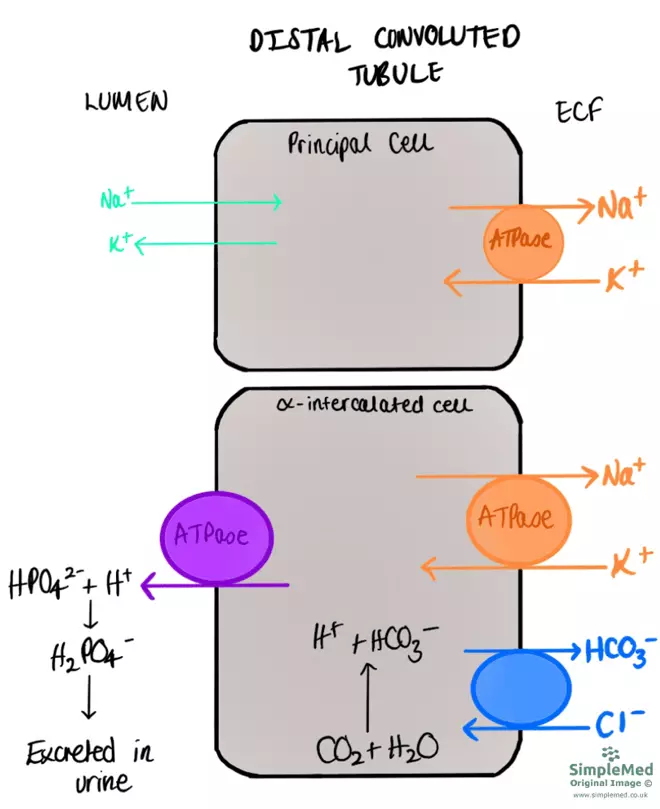
Diagram - The movement of phosphate ions out of α-intercalated cells in the distal convoluted tubule
SimpleMed original by Dr. Peter Parkinson
When there is an increase in the pCO2 (for example secondary to hypoventilation), the concentration of H+ ions secreted into the urine will increase, causing the pH of the blood to decrease. The kidneys will begin to excrete less HCO3- ions, so there are more HCO3- ions present in the blood. This will help the pH to go back to normal, and is a form of compensation (see later).
If the pCO2 begins to decrease (for example secondary to hyperventilation), the pH of the plasma will begin to increase. The kidneys will then excrete more HCO3- ions to try to bring the pH of the plasma back within normal range. However, this change can take quite a long time, so any compensation (change in HCO3-) seen in these patients is a chronic change rather than an acute one.
Potassium Ions and Acid Base Disturbances
There is a relationship between potassium ions and the plasma pH.
To regulate the amount of H+ in the plasma, the body can move it in or out of cells in exchange for K+ ions. This means that any changes in plasma pH can cause changes in K+ levels. This swap is known as the reciprocal cation shift.
If the pH of the plasma is acidotic, the body will try to move more H+ ions into cells to lower the plasma pH and this results in more K+ ions being transported into the blood from cells. There will also be reduced excretion of K+ ions in the distal nephrons. Overall, acidosis will lead to hyperkalaemia.
If the pH of the plasma is alkalotic, H+ ions will be transported out of cells to raise plasma pH, as K+ ions are moved into cells. The kidneys will excrete more K+ ions to the urine. Overall, alkalosis will lead to hypokalaemia.
This reciprocal cation shift can also be put into practice where the potassium abnormalities come before the pH changes:
- If a patient develops hyperkalaemia, K+ ions will be moved into cells in exchange for H+ ions which will move out of the cells. This means that the pH inside the cells of the nephron will become more alkaline. This then favours increased HCO3- excretion into the urine. Overall, a patient will develop metabolic acidosis as a result of their hyperkalaemia.
- If a patient develops hypokalaemia, K+ ions will move out of cells in exchange for H+ ions which will move into the cells. This means that the pH inside the cells of the nephron will become more acidic. This then favours HCO3- recovery from the filtrate (urine) and increased excretion of H+ ions into the urine. Overall, a patient will develop metabolic alkalosis as a result of their hypokalaemia.
Diabetic ketoacidosis (DKA) is a key condition when considering the relationship between pH and potassium disturbance: it causes both metabolic acidosis and systemic depletion of potassium ions. In DKA, ketones are produced as a source of fuel, and because ketones are acidic, this causes K+ ions to move out of the cells as blood pH falls due to increased ketoacid levels and reduced insulin, which normally drives potassium into cells. However, with there being increased concentration of glucose present in the lumen of nephrons, this increases the osmolality and K+ ions are trapped in the lumen as well. This results in increased concentration of K+ ions being excreted in urine and a depletion systemically.
Compensation and The Anion Gap
As previously discussed, the plasma pH depends on the ratio between concentration of hydrogen carbonate ions and the partial pressure of carbon dioxide.
If there are changes in the pCO2, the kidney can compensate by alternating the excretion of HCO3- ions. In respiratory acidosis, the kidneys will compensate by excreting less HCO3- ions and in respiratory alkalosis, the kidneys will compensate by excreting more HCO3- ions. However, it can take the kidneys several days to compensate, so this is not an immediate management strategy.
In a patient with compensated respiratory acidosis, the arterial blood gas will show: high pCO2, raised [HCO3-] with a relatively normal plasma pH.
In a patient with compensated respiratory alkalosis, the arterial blood gas will show: low pCO2, low [HCO3-] with a relatively normal plasma pH.
The anion gap is the difference between the concentrations of cations and anions in the blood. It includes the concentrations of sodium and potassium ions as the cations, and the concentrations of chloride and hydrogen carbonate ions as the anions.
The normal anion gap is usually around 8 to 12 mmol/L when potassium is excluded from the calculation (local reference ranges may vary). This reflects the presence of unmeasured anions in normal plasma.
It can be helpful to use the anion gap to work out the cause of an abnormality, for example:
- Changes in potassium concentration have little direct effect on the anion gap, as potassium contributes minimally to the calculation.
- In compensated respiratory acidosis, renal compensation leads to a raised HCO3- concentration, and the anion gap is typically unchanged.
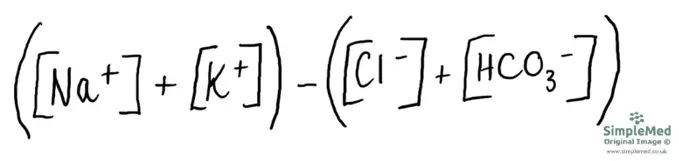 Image - The anion gap equation
Image - The anion gap equation
SimpleMed original by Dr. Peter Parkinson
It is important to remember that the anion gap is calculated using a limited number of measured ions, but it does reflect the build-up of unmeasured anions such as lactate or ketoacids, which increase the gap.
The anion gap is increased if HCO3- ions are replaced by another anion; this is seen when there is metabolic production of an acid such as diabetic ketoacidosis and lactic acidosis.
With a renal cause of metabolic acidosis, the anion gap can be normal. During the excretion of HCO3- ions, they are replaced by chloride ions in a 1:1 ratio, meaning the anion gap will be unaffected.
In metabolic alkalosis, the kidneys try to compensate by reducing H+ ion excretion and HCO3- recovery. Metabolic alkalosis is also accompanied by systemic volume depletion, this leads to the kidneys increasing the recovery of sodium ions and HCO3- ions whilst excreting H+ ions. This means that the resorption of anions and cations is balanced, so the anion gap will not change.
Arterial Blood Gas Interpretation
As mentioned at the start of this article, all of this information is provided on an arterial blood gas (ABG). It is important to remember that pCO2 and HCO3- results can be outside the normal range due to pathology relating to their formation or removal, or because they have compensated for a change in the other variable to get the pH closer to the normal range.
This is one method that can be used to interpret the results of an ABG:
First look at the pH - is it low (below 7.35) indicating acidosis, or high (over 7.45) indicating alkalosis?
If Acidotic:
- Look at the pCO2 - is it high (respiratory cause of the acidosis) or low (compensating for a metabolic cause)
- Look at the HCO3- - is it low (metabolic cause of the acidosis) or high (compensating for a respiratory cause)
If alkalotic:
- Look at the pCO2 - is it low (respiratory cause of the alkalosis) or high (compensating for a metabolic cause)
- Look at the HCO3- - is it high (metabolic cause of the alkalosis) or low (compensating for a respiratory cause)
Things to remember with ABGs:
- Remember that changes in HCO3- reflect renal handling and metabolic processes. Renal compensation takes time, but a low bicarbonate does not by itself indicate whether a disorder is acute or chronic.
- Remember that compensation is done to get the pH back into the normal range. This means that there are two levels of compensation:
- Partial compensation occurs when both variables are outside the normal range, and the pH has not been pushed back into the normal range.
- Full compensation occurs when both variables are outside the normal range, but the pH is within normal range.
- It is always important to interpret the ABG in the clinical context of the patient - this may help determine whether the cause is chronic or acute.
Edited by: Dr. Maddie Swannack
Reviewed by: Dr. Thomas Burnell
- 7528

