By Dr. Marcus Judge and Dr. Ethan Kavanagh
Next Lesson - Gram Staining and Infection Investigations
Abstract
- Antigen-presenting cells activate different T-lymphocytes and B-lymphocytes to help fight against invaders
- MHC is a series of proteins that the adaptive/acquired immune response needs to recognise foreign material
- Naïve T-cells differentiate into cytotoxic T-cells and several T-helper cells (Th1, Th2, Th17) which have different responses depending on the current active infection
Core
To understand this article, we advise that you read the Innate Immune Response, or have a rough understanding of it.
As outlined in the previous article although the innate response is rapid, it lacks memory and the ability to change the intensity of a response; this is where the adaptive immune system comes in. So why does the adaptive immune response not start immediately fighting bacteria on infection?
While the innate immune response is coordinating the early ‘front line’ response against the invaders, the adaptive response must wait for its cells to become activated. Certain types of cells, called antigen presenting cells (APCs), are found throughout the body where they sample their environment for foreign antigens. APCs are a diverse group including macrophages, monocytes (macrophages in the blood), dendritic cells, Langerhans cells, and B-lymphocytes.
An antigen presenting cell, e.g. a macrophage, will phagocytose a foreign pathogen, as part of the innate immune response. After taking in the pathogen and degrading it within lysosomes, a small sample of the antigen from the pathogen is then displayed ("presented") (hence the name antigen presenting cell) on the surface of the macrophage using a molecule called a Major Histocompatibility Complex (MHC). This process can be broken down into 3 simple steps of: Capture (phagocytosis), Processing (digestion down to antigen fragments) and Presentation (using an MHC).
The next step is T cell activation. This occurs with binding between the antigen-displaying MHC on the macrophage surface and a T-lymphocyte. This requirement for specific antigen recognition by the T cell receptor, which causes a delay in activation of the adaptive immune response. Once the T-lymphocytes are activated, both APC’s and a T-lymphocyte are required to activate B-cells to produce specific antibodies, signalling using cytokines. T lymphocytes also begin to proliferate and starting to mount their own attack against the invading pathogen. We’ll come back to T- and B-lymphocytes, as we need to discuss how the APCs do their job.
Presentation of antigens is classified by the major histocompatibility complex. The MHC consists of a set of cell surface proteins that are split into two main classes (there is a third, but it isn’t as important as the first two). In humans, the MHC class is based on the human leukocyte antigens (HLAs) it expresses.
There are two main classes:
MHC class I- molecules are expressed on all cells not just APC’s and express HLA-A, HLA-B and HLA-C. MHC Class I molecules bind to T-lymphocytes (specifically cytotoxic T-cells, aka T-killer cells) via CD8+ receptors. Only intracellular microbes (e.g. a virus which has entered the cell) are presented via MHC class I molecules.
MHC class II- molecules are only found on antigen presenting cells. This is as MHC Class II is used to present extracellular microbe antigens. APC’s are the only cells which can carry out phagocytosis to obtain the antigens from extracellular microbes, so must be the only cells with MHC class II proteins. Once an APC processes the antigen, it puts a little piece of it (called the epitope) on its cell surface along with a class II molecule. These APCs can then bind to the CD4+ receptors on naïve T cells (those that have matured from the thymus but not been activated before i.e. bacteria the body has not fought before) which differentiate into T helper (Th) cells. There are different types of T-helper cells - what determines the fate of each naïve T-cell are the co-stimulatory proteins (cytokines) that also bind to the naïve T-cell. The three most important types of T helper cell are Th1, Th2, and Th17. Extracellular molecules taken up by antigen presenting cells are presented via MHC class II molecules.
Key facts about MHCs:
- The MHC molecules have a broad specificity so that a single cell can present multiple antigens from the same microbe to increase the immune response.
- The specific HLA molecules are present on a person's MHCs determines their susceptibility to certain infections e.g. HIV. Some HIV positive individuals have a specific set of HLA molecules which mean they have a slowly progressing disease as the T-cell response is effective. This is as these HLA molecules present key viral peptides which are crucial in virus survival. Those who only have HLA’s which present non crucial peptides (which may as a result mutate in the virus) as a result have a poorer T-cell response and rapidly progress).
- A mismatch in MHC molecules between a donor and recipient in an organ transplant can cause rejection of the organ. If the organ transplanted is bone marrow, a mismatch in MHC molecules can result in Graft vs Host disease whereby the new white blood cells produced by the donor marrow attack the host’s tissues.
- Having certain MHC molecules can predispose individuals to certain autoimmune conditions e.g. insulin-dependent diabetes mellitus.
A helpful way of remembering which class of MHC binds to which CD receptor is:
When you multiply the MHC class and CD number together you should get 8:
MHC Class I binds to CD8+ (1x8)
MHC Class II binds to CD4+ (2x4)
Fighting off intracellular microbes - MHC Class 1
An intracellular organism, for example a virus invading a cell to take over its replication machinery, can infect any nucleated cell. For this reason, MHC class I is expressed on all nucleated cells.
An APC can present antigens on both MHC class I and MHC class II molecules. Cross-presentation specifically refers to the ability of some APCs to present extracellular (phagocytosed) antigens on MHC class I, allowing activation of CD8+ T cells.
So, if an APC (e.g. a macrophage) with its epitope (the part of an antigen molecule to which an antibody attaches itself) bound to an MHC molecule arrives at a naïve T-cell -what next?
If that APC happens to be expressing the epitope in an MHC class I molecule, this will be bound by a T cell expressing the CD8+ receptor, leading to the activation of a cytotoxic T-cell. This ‘killer’ cell then kills any infected body cells.
Fighting off extracellular microbes - MHC Class 2
Once a MHC class II molecule binds to the CD4+ receptor on T-cells, they can differentiate into Th1, Th2, or Th17 cells.
Th17 just has one action we need to know, and that is activating neutrophils by interleukin (IL) 17.
Th2 cells have a few different responses:
- Increases the number of eosinophils to help kill parasites (via IL-5)
- Activates B-cells to produce antibodies (for phagocytosis) and complement (via IL-4)
- Activates mast cells which are responsible for local inflammation and our allergic reactions (via IL-4)
A Quick Summary so far
Cell-mediated immunity:
- This fights off intracellular microbes such as viruses, bacteria, and protozoa
- Cell-mediated immunity is an immune response that does not involve antibodies, but rather involves the activation of phagocytes, antigen-specific cytotoxic T-lymphocytes, and the release of various cytokines in response to an antigen
- The receptors and cells involved are CD4+ differentiating naïve T-cells to Th1 cells and CD8+ differentiating naïve T-cells into cytotoxic T-cells
- Attack mechanisms are macrophages and cytotoxic T cells
Humoral immunity:
- This fights off extracellular microbes such as fungi, bacteria, parasites, and worms
- Humoral immunity is the aspect of immunity that is mediated by macromolecules found in extracellular fluids such as secreted antibodies, complement proteins, and certain antimicrobial peptides
- The receptors and cells involved are CD4+ receptors differentiating T-cells into Th2 and Th17 cells
- Attack mechanisms are antibodies, complement, and phagocytosis
Primary and Secondary Immune Antibody-based Response
If you are unlucky enough to have had chicken pox, the good news is you shouldn’t ever get it again. This is because if Varicella zoster has invaded before, the body is now able to destroy it before it causes disease or symptoms (the secondary immune response), since it has developed a memory against it.
The primary response is your body’s first response to a new pathogen that it has never encountered before. The primary antibody produced against this response is IgM, which is less specific than an IgG reaction. It takes around 5-10 days for a peak primary immune response to be mounted, which is slow.
On the second exposure to the same pathogen, the secondary immune response is activated. This is primarily IgG based and takes less time - usually 3-5 days to reach peak. This is a much faster and stronger response which also lasts longer than the primary immune response with IgM. This is also the basis behind vaccines where the initial exposure is to an antigen or an attenuated or dead pathogen so that the memory can be built allowing for a secondary immune response to be mounted immediately when the pathogen is first naturally encountered. This is beneficial as some diseases e.g. Polio cause death and disability even after one exposure. The use of vaccines means that this unneeded suffering is not required to build up a secondary response.
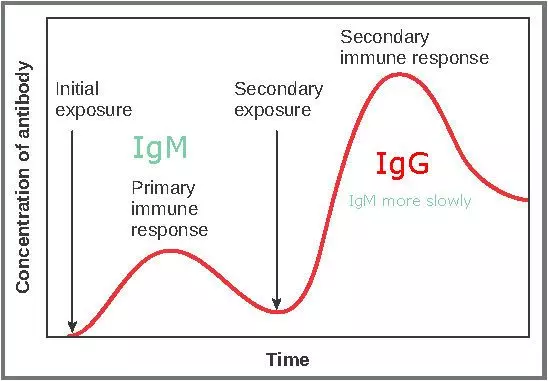
Figure: Shows the response of the adaptive immune system to a standard infection. The primary response on first exposure will be mainly IgM mediated. The secondary response on second exposure will be IgG (memory cells) as these activate more quickly than IgM. You will still get an IgM response in the secondary immune response
Creative commons source by OpenStax College, edited by Dr. Marcus Judge [CC BY 3.0 (https://creativecommons.org/licenses/by/3.0)]
The important thing to remember is that you won’t get a secondary immune response with the same pathogen unless the same antigens that were there during the primary immune response are present. An example where this is relevant is with the influenza virus. It has certain antigens, such as haemagglutinin and neuraminidase (which are also used to name certain flu strains, eg. H1N1), which are slightly different between flu strains. It is possible to get the flu annually because the antigens are constantly mutating and memory is not for the new unencountered strain. This means the flu vaccine is also only effective for that specific season as any new mutated strains cannot be anticipated.
Edited by: Dr. Ben Appleby and Dr. Marcus Judge
- 12542

