Next Lesson - Thrombosis and Embolism
Abstract
- Haemostasis is the physiological process that stops bleeding at the site of vessel injury while maintaining normal blood flow elsewhere.
- The process allows for blood clots to form via the clotting cascade in order to limit damage to the body and prevent excessive bleeding.
- Clotting factor function can be measured by the PT and APTT tests; platelet function has historically been assessed using bleeding time, although this is now rarely used in clinical practice.
- Clotting disorders can be inherited e.g. haemophilia or von Willebrand disease, or acquired e.g. DIC.
- Thrombocytopenia can lead to petechiae, and thrombophilias increase the chance of clot formation.
Core
Haemostasis (not to be confused with homeostasis) comes from haemo (pertaining to blood) and stasis (to halt) and thus literally translates to halting blood. It is an essential process in the moments after vessel injury that prevents small wounds from becoming fatal.
The Three Steps of Haemostasis
- Vessel wall damage
- The severed or damaged artery contracts. This contraction isn’t enough to stop the bleeding but does assist in reducing the pressure ‘downstream’ of the site of injury and control the bleeding. Contraction is less pronounced in veins due to their thinner smooth muscle layer; however, bleeding is usually less severe because venous blood flow is under lower pressure.
- A primary haemostatic plug of activated platelets will form at the site of injury. This plug will stick to the injured vessel and the tissue outside of it. Once more, this won’t stop the bleeding completely but will help to control the bleeding. The primary plug will form seconds to minutes following the vessel severing.
- A secondary haemostatic plug forms as fibrin filaments stabilise the fragile platelet plug to form a blood clot. This will take around 30 minutes to form in a healthy individual.
Below are the aspects of haemostasis and clotting in more detail.
Platelets are cell fragments which have no nucleus and are involved in the process of blood clotting. There are approximately 150-400 x109 cells per litre of blood. Platelets are formed in the bone marrow by the process of budding from the cytoplasm of megakaryocytes.
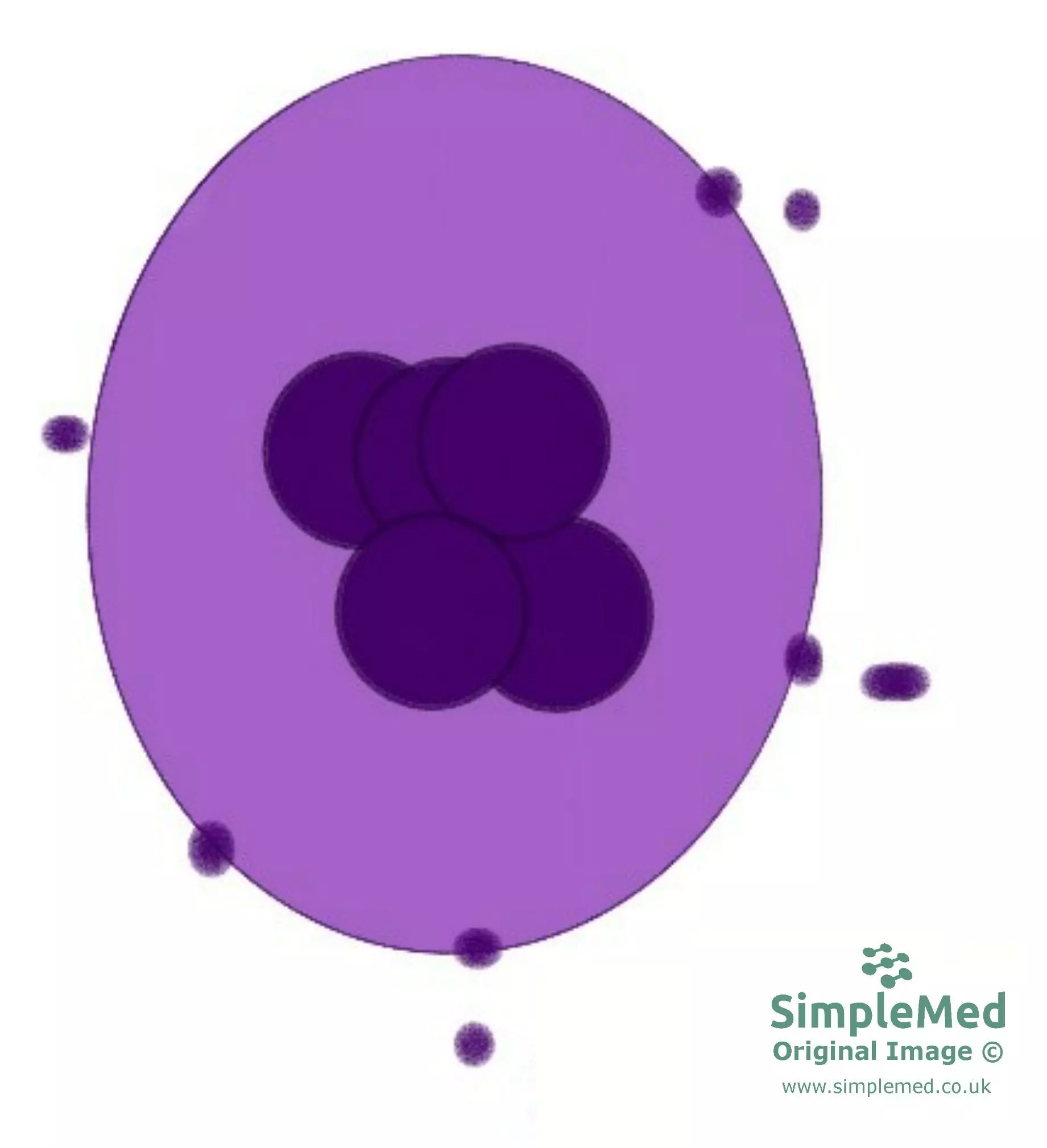
Image - Platelets (represented by the small dark purple circles) budding from a megakaryocyte
SimpleMed original by Benjamin Norris
Platelets can be activated in numerous ways:
- Contact with collagen surfaces - collagen is found in the blood vessel wall below the endothelium and is therefore exposed when the blood vessel is damaged and can activate platelets.
- ADP - released by activated platelets and red blood cells. This causes an amplified platelet response.
- Thromboxane A2 - a powerful platelet aggregator that is released by activated platelets in order to amplify the platelet response.
- Thrombin - activated in the clotting cascade and recruits more platelets.
Once activated, platelets will adhere to von Willebrand factor (vWF), which is concentrated on the subendothelial basement membrane of blood vessels. They will then proceed to aggregate with other platelets, growing the haemostatic plug. The platelets are cross linked by a clotting factor called fibrin formed in the clotting cascade (explained in more detail below) which will form a mesh structure to reinforce the platelet plug. Once a platelet is activated, it will secrete factors which recruit more platelets such as ADP and thromboxane A2.
The drug aspirin irreversibly deactivates cyclooxygenase, an enzyme responsible for thromboxane A2 production. Therefore, long-term low-dose aspirin is used as secondary prevention against myocardial infarction and stroke (i.e. aspirin makes it more difficult for blood clots to form).
For blood to clot, fibrin is needed which are threads of protein that can cross-link to form a fibrin mesh. This mesh catches platelets and holds the blood clot together to strengthen the platelet plug. The formation of fibrin is achieved via the clotting cascade, which is shown in the diagram below.
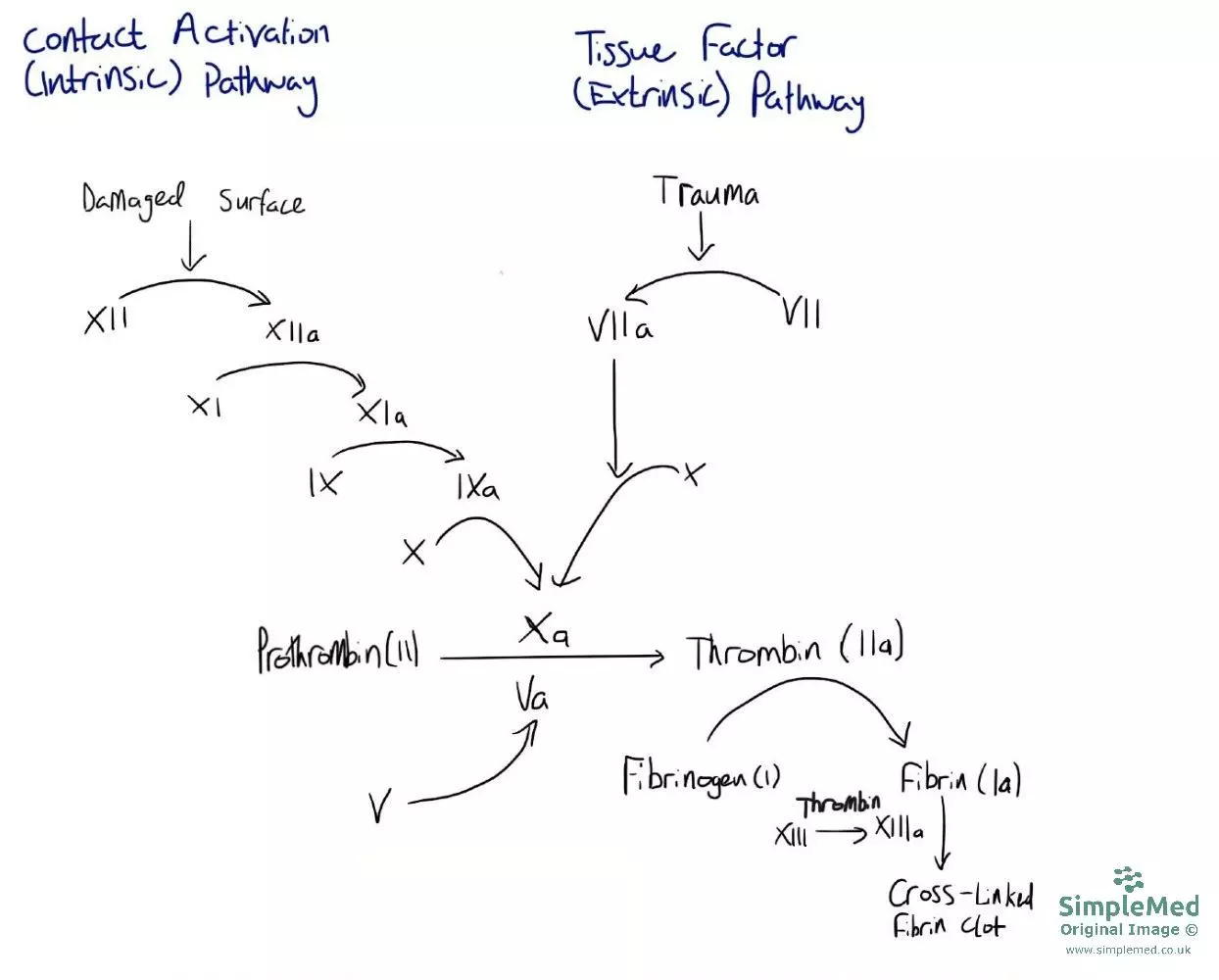
Diagram - The clotting cascade. It can be activated by two different pathways: the intrinsic and extrinsic pathways. The two pathways come to the same result which is the formation of fibrin from fibrinogen used to cross-link platelets and form the platelet plug
SimpleMed original by Benjamin Norris
Coagulation can occur via two pathways which produce the same result. The two pathways use some different clotting factors shown in the Venn diagram. This means that timings of these pathways can be measured when investigating coagulation disorders to narrow down the clotting factors affected. The two pathways and their measurement are explained below:
- Intrinsic pathway (measured by APTT) - Triggered by the exposure of collagen after a blood vessel is damaged.
- Extrinsic pathway (measured by PT) - Triggered by the release of thromboplastin from damaged endothelial cells. It can be released in response to breaking of the blood vessel wall, hypoxia, sepsis, malignancy and inflammation.
PT can also be expressed by the international normalised ratio (INR) which compares the PT to a normal value in the form of a ratio. For example, an INR of 1 is normal, and blood with an INR of 3 will clot more slowly, for example if a patient is on warfarin.
There is an additional measurement which has historically been used to investigate clotting by measuring how long the patient bleeds for after a break to the skin. A longer bleeding time (BT) suggests a problem with platelet aggregation, although BT is now largely obsolete and has been replaced by laboratory platelet function tests.
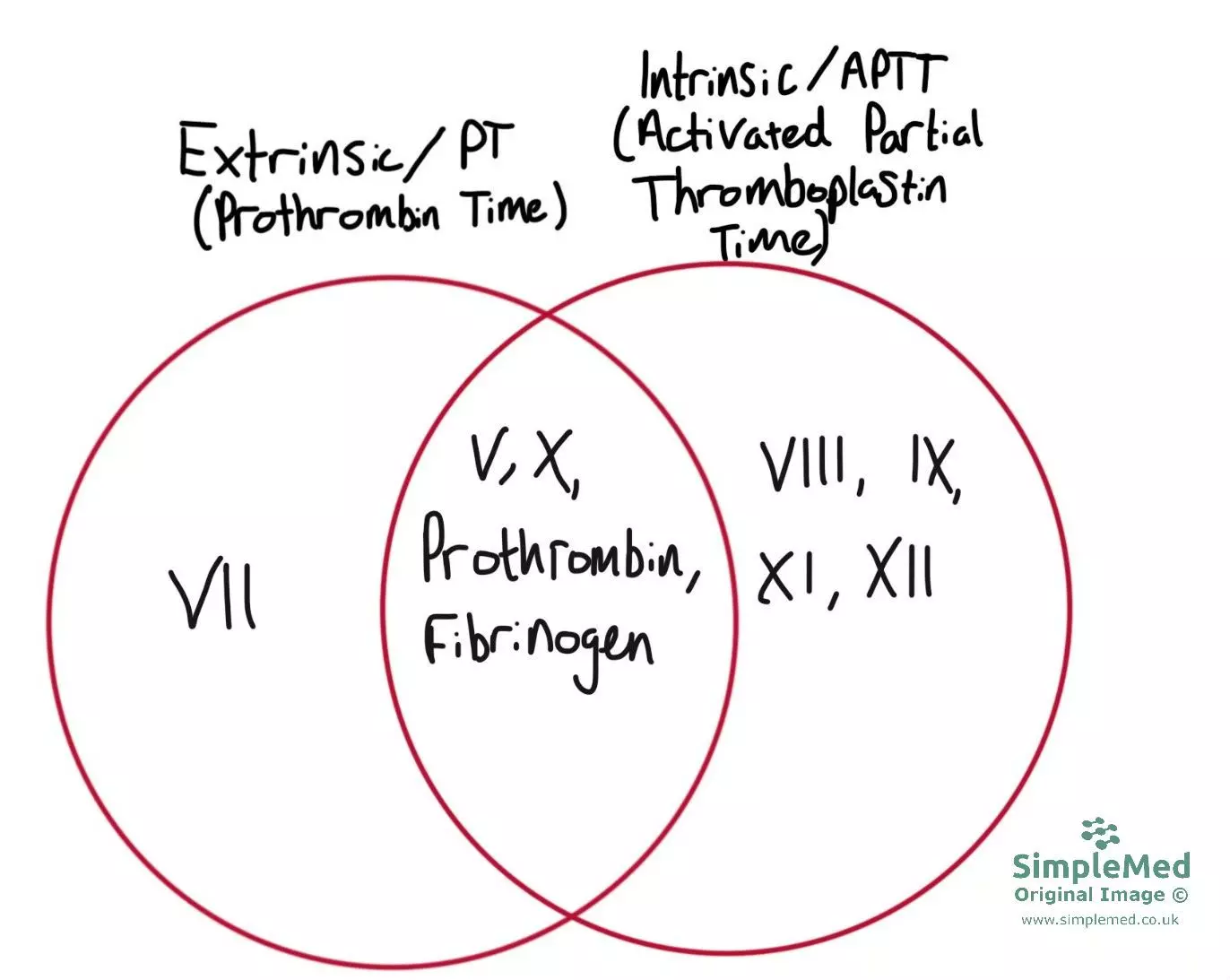
Diagram - A Venn diagram illustrating the factors present in the extrinsic and intrinsic pathways
SimpleMed original by Benjamin Norris
These factors include dilution of clotting factors by blood flow and natural anticoagulants. The main natural anticoagulants in the blood are antithrombin III, protein S and protein C. Natural anticoagulants don’t destroy clots (that deed is performed by the enzyme plasmin in fibrinolysis); rather they oppose the formation of fibrin in the first place to control the clotting cascade.
As platelets within the clot contract, they pull on fibrin via actin-myosin filaments. As a result of this, the edges of the wound to which the clot has adhered are drawn together to a certain extent. This is wound contraction, a part of the healing process of the blood vessel.
The endothelium secretes a substance known as plasminogen which is converted to plasmin by tissue plasminogen activator (tPA). The activity of tPA is increased by the presence of fibrin. Plasmin is the major enzyme of the fibrinolytic system and destroys clots by degrading fibrin. Fibrin degradation/split products released in this process also inhibit clotting. This system ensures that clots do not persist beyond the time for which they are needed.
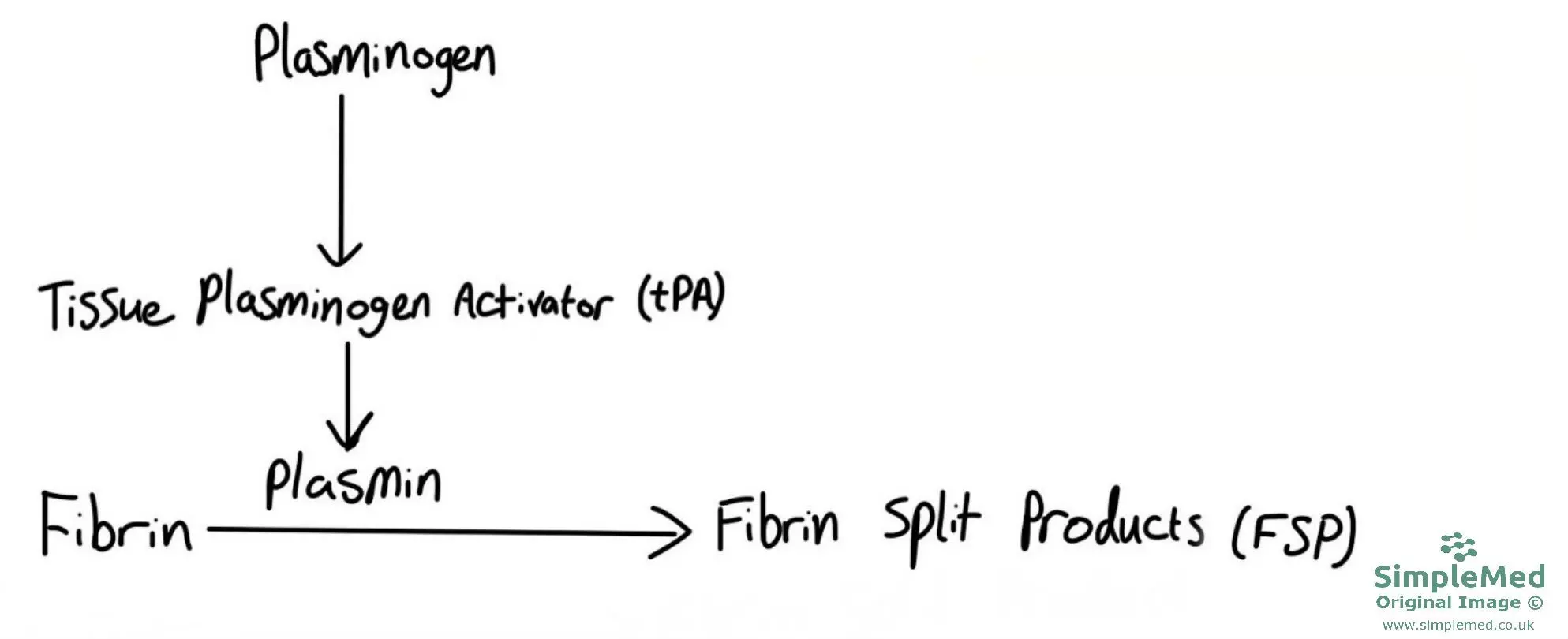
Diagram - The factors involved in fibrinolysis. NB Fibrin Split Products (FSPs) can also be referred to as Fibrin Degradation Products (FDPs)
SimpleMed original by Benjamin Norris
tPA can be used therapeutically in dissolving thrombi and thromboembolisms, however, a side effect of this can be undesired bleeding. An additional fibrinolytic agent which can be used therapeutically is streptokinase, however this is obtained from streptococci, meaning that it is antigenic and can only be used once.
Disseminated Intravascular Coagulation (DIC)
Disseminated intravascular coagulation is a pathological activation of the coagulation cascade. In DIC, clotting activators enter the systemic circulation and many microthrombi (small pathological clots in the vessels) are formed throughout the circulation. During this process many coagulation factors and platelets are consumed.
When enough of these clotting factors have been used, haemorrhage can begin to occur as the body has no more platelets or clotting factors to form clots when they're actually needed. The affected patient is at risk of bleeding, neurological impairment, gangrene, renal failure, respiratory distress and a wide variety of other complications. Red blood cells are often damaged in DIC as they are sheared by flowing past the microthrombi, and between this and haemorrhage the patient can develop anaemia.
Triggers for DIC can include malignancy, massive trauma (e.g. burns), infections, massive haemorrhage, and transfusion. Although in the short term DIC can be managed by transfusing the patient with clotting factors and platelets, in order to truly treat the condition, the underlying cause must be treated first.
There are two main types of haemophilia, both of which are congenital disorders of clotting factors. These two types are haemophilia A and haemophilia B (also known as Christmas disease). Platelets are unaffected in haemophilia, however, as there are inadequate amounts of clotting factors, less fibrin is produced.
Both varieties of haemophilia are X-linked recessive and are therefore much more common in males than in females. Haemophilia A and haemophilia B are clinically indistinguishable from one another, with both having normal bleeding time and PT but prolonged APTT (factor VIII and IX are used in the intrinsic pathway causing the APTT to be prolonged, but neither factor is involved in the extrinsic pathway so PT is normal).
Haemophilia A is either a deficiency of the amount of factor VIII, or reduced factor VIII activity It is the most common hereditary disorder associated with serious bleeding.
Individuals with mild haemophilia A may experience prolonged bleeding after major trauma or surgery, and after venepuncture. In more severe cases, patients can present with spontaneous bleeding into joints (haemarthrosis), intramuscular haemorrhage and, in severe cases, intracranial haemorrhage.
Mild haemophilia A is seen with 5-40% of normal factor VIII activity, moderate disease with 1-5%, and the severe form with under 1% of factor VIII.
Patients with haemophilia A can be managed through infusion of recombinant factor VIII.
Haemophilia B is a deficiency in factor IX, and presents with similar symptoms to haemophilia A, but with an increased risk of bruising and experiencing mucosal bleeds. It can be treated through an infusion of recombinant factor IX.
Von Willebrand disease is the most common inherited bleeding disorder and can range in nature from asymptomatic to severe. Von Willebrand factor is either deficient or abnormal in this condition.
The function of this protein includes protecting factor VIII from premature destruction and assisting in the formation of the platelet plug. Both bleeding time and APTT can be affected by this condition.
Features include bleeding following minor trauma or surgery, epistaxis and menorrhagia. Bleeding into muscles and joints is rare.
Management depends on the type and severity of von Willebrand disease. Many patients respond to desmopressin, while others require concentrates containing von Willebrand factor (with factor VIII).
Thrombocytopenia is a condition in which the platelet count is less than 100 x109/L. In this condition the bleeding time is affected, but the PT and APTT remain unaffected, as these are an indicator of clotting cascade function.
Patients with thrombocytopenia can present with petechiae, which are small red or purple spots under the skin; these occur due to spontaneous bleeding from capillaries.
Thrombocytopenia has a variety of causes such as autoimmune thrombocytopenic purpura, DIC, liver failure or drugs such as heparin.
Thrombophilia can be due to congenital or acquired defects of haemostasis that lead to an increased chance of thrombosis (pathological blood clotting).
Examples of thrombophilias include factor V Leiden thrombophilia, antithrombin deficiency, protein S or protein C deficiency, and antiphospholipid syndrome.
Edited by: Dr. Bethany Turner
Reviewed by: Dr. Marcus Judge
- 13091

