By Dr. Ethan Kavanagh and Dr. Marcus Judge
All done here! Check out our other subjects!
Abstract
- Travel-related diseases are often rare within Western countries. Travel to an area in which they are common can make a diagnosis considerably more likely.
- Malaria is a deadly protozoal infection by a Plasmodium species. It is spread by Anopheles mosquitoes and is diagnosed using blood films.
- Legionnaire's disease is a bacterial infection by Legionella species leading to respiratory-related symptoms. It is often caught from poorly cleaned water sources including showers and air conditioning systems.
- Dengue fever, schistosomiasis and typhoid fever are all common types of travel-related infections also known as Neglected Tropical Diseases with different signs/ symptoms and diagnosis criteria.
Core
What are Travel-related Infections?
Travel related infections occur when patients have travelled to another location which has a different set of common diseases - usually a location with a considerable climate difference or within the tropics. Some of the diseases acquired are referred to as NTDs (Neglected Tropical Diseases), due to their high impact in low income and poor communities despite being mostly treatable.
These diseases usually require a vector (transmission vessel) that is indigenous to the country or area that they are located within e.g. the female Anopheles mosquito, which is the vector for the Plasmodium species that cause malaria, is not present in the UK but is common in sub Saharan Africa.
Many travel-related infections should be identified and treated promptly, as they can be life-threatening. Some are transmissible between people, while others are acquired via vectors or environmental exposure, so transmission risk depends on the specific infection.
If a tropical disease is suspected, a comprehensive travel history should be taken. As it may not be readily apparent to the patient that their illness is related to their travel (some cases of malaria may have an incubation period of close to a year), patients are unlikely to tell you about their travels as they are not seem relevant. This can also mean easily treatable tropical infections can have time to develop complications as they are not considered in a differential diagnosis. This delay in diagnosis is of further concern if the particular disease is contagious. Therefore a good travel history can prevent the disease from; developing complications, spreading throughout the hospital or community and, allows for adequate containment and treatment to be set up.
It is important to be precise when establishing a travel history as some countries may have different strains of the same microorganism whose prevalence may vary by time of year. These different strains may also have differences in antibiotic or drug resistance, therefore this information is key in guiding treatment.
There are a number of travel-related infections, but only a few of the most important ones to be aware of will be covered here. Where the patients treated have travelled to recently will determine which infections are more likely than others (i.e. the most prevalent for a particular area).
Malaria is one of the most common and deadly infections in the world, spread rapidly by mosquitoes.
Malaria involves infection of the patient's red blood cells (erythrocytes) with protozoal Plasmodium microorganisms, which then multiply within and destroy the erythrocytes, causing severe sickness, anaemia, fever of a cyclical nature and potentially death. Malaria can be identified on a patient's blood film, where parasites may be seen within infected red blood cells. Microscopy remains important, often alongside rapid diagnostic tests, to confirm the diagnosis.
Presentation
Recognising malaria is vital in a patient that has a travel history in areas that have a high prevalence of malaria, such as South and Central America, Oceania, Africa, the Caribbean, Middle East and Southeast Asia. A presenting patient with suspected malaria might present with:
- Tachycardia
- Hypotension in severe cases
- Reduced oxygen saturation in severe disease
- Mild confusion
- History of mosquito bites
- Hepatosplenomegaly
- Vomiting and nausea
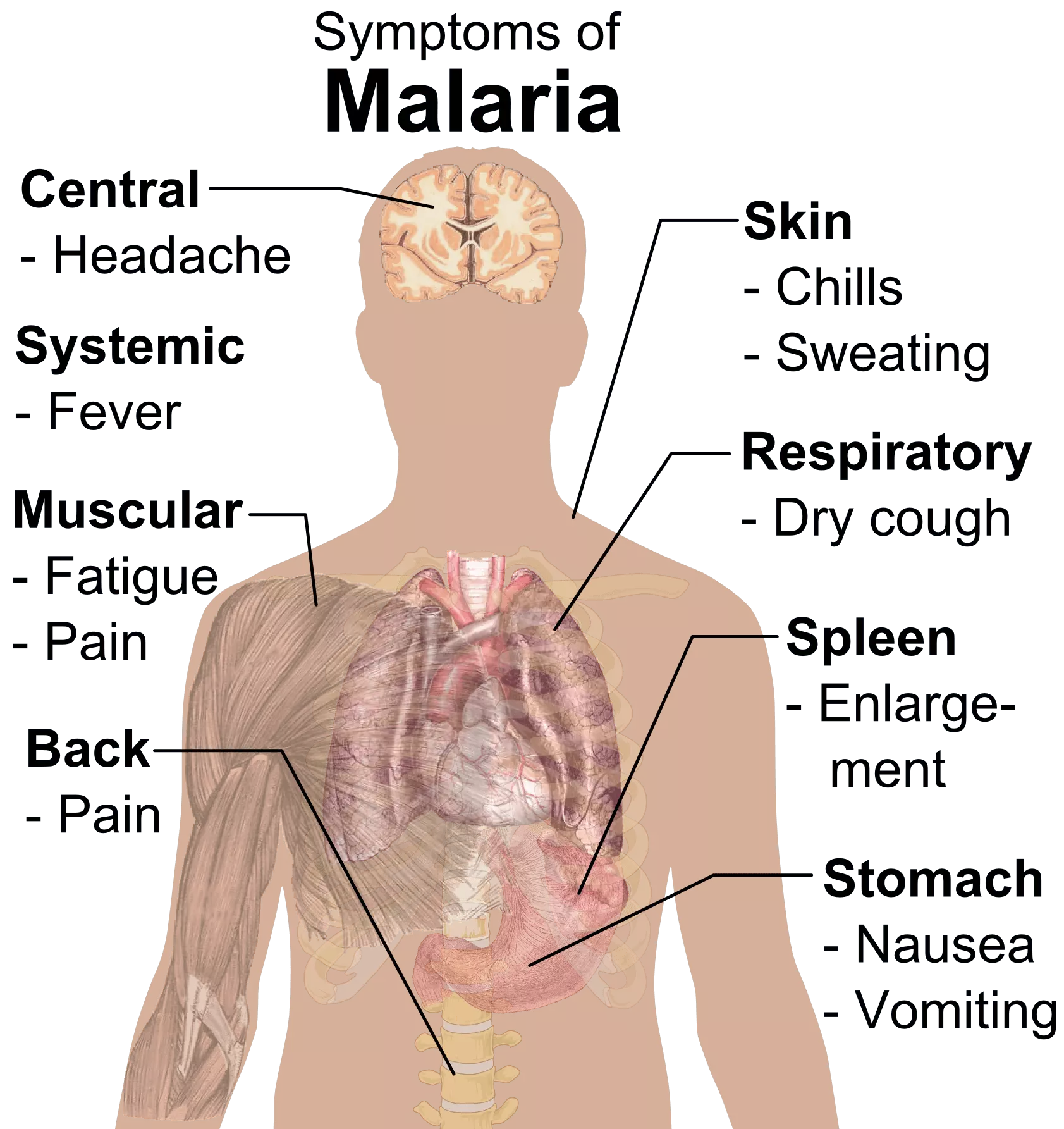
Diagram: The systemic symptoms of Malaria - any of these symptoms after a travel history to a high-risk area should be suspected of being caused by Malaria.
Creative commons source by [Häggström, Mikael (2014). "Medical gallery of Mikael Häggström 2014". WikiJournal of Medicine 1 (2). DOI:10.15347/wjm/2014.008. ISSN 2002-4436. Public Domain.orBy Mikael Häggström, used with permission. [Public domain]]
These symptoms are also easily indicative of bacterial sepsis - as such, a good history is key to understanding a patient's presentation.
Pathophysiology
Malaria is caused by different species of the protozoal genus Plasmodium. There are many different species, but only five actually cause malaria in humans:
- Falciparum (responsible for approximately 50% of malaria cases and is the deadliest human parasite, found on all continents bar Europe but most prevalent in Africa)
- Ovale
- Vivax
- Malariae
- Knowlesi
Regardless of strain, malaria is carried by the female Anopheles mosquito (the mosquito is the vector). Once a person is infected, the malaria then has an incubation period inside the patient where it begins to replicate so that it can be passed on from the host. In the case of malaria, this could be anywhere from six days to over a year!
Each strain has a slightly different life-cycle when infecting humans, but we will focus on P. falciparum's life-cycle, the most common strain of malaria.
Once a mosquito bites someone with its proboscis, the plasmodium moves from the mosquito’s salivary gland and into the blood stream of the person the mosquito has attacked. When it is in the bloodstream, the plasmodium is in the sporozoite stage of its life cycle. Once these sporozoites get into the bloodstream, they go straight for the liver and get inside the liver parenchyma (functional tissue).
The P. falciparum then begin to multiply asexually (still in the liver, but rapidly) and mature from sporozoites to merozoites. Once they’re ready, they burst out from the liver and go straight into the bloodstream where they attack and enter erythrocytes. P. falciparum can invade erythrocytes of all ages.
Now that they’ve gotten into the erythrocyte, the plasmodium begins to replicate (asexually) and mature (over about 2-3 days). When they’re ready to burst out of the erythrocyte, the erythrocyte is called a schizont. What actually bursts out is a mass of merozoites, ready to continue the cycle by infecting and destroying even more erythrocytes.
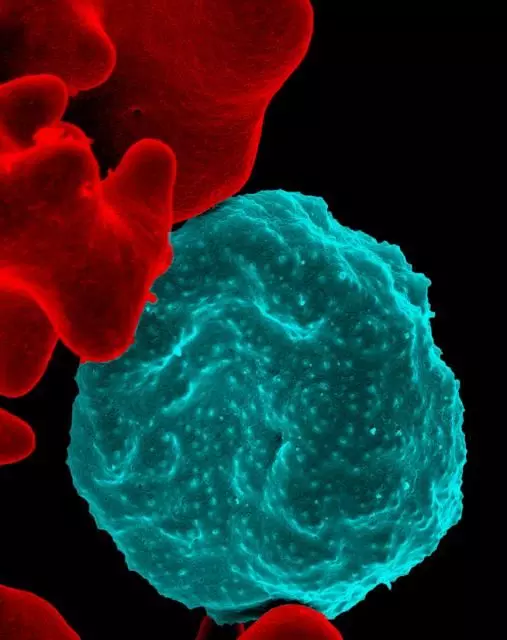
Image: Colourised scanning electron micrograph of red blood cell infected with malaria parasites, which are colourised in blue. The infected cell is in the centre of the image area. To the left are uninfected cells with a smooth red surface.
Creative commons source by NIAID [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)]
Some plasmodia don’t follow the same path as the others once inside the erythrocytes and become gametocytes instead. These gametocytes stay inside the erythrocyte (the erythrocyte doesn’t rupture) and are instead sucked up by a different mosquito that is feeding on the same human. The infected erythrocyte then makes its way to the mosquito’s gut, forms a zygote, matures inside the mosquito by sexual reproduction and releases a mass of sporozoites. These sporozoites then travel to the mosquito’s salivary gland, ready for the mosquito to feed on someone else.
The key takeaway message is that malaria invades and either destroys red blood cells, causing many of the symptoms seen, or remains within cells to be taken back up by other mosquitoes.
Incubation time is the time from when a person becomes infected until they start to see the onset of symptoms. For malaria, this can range dramatically from six days (P. falciparum) to a few weeks (P. malariae). Antimalarial drugs can also delay malaria symptoms until long after the patient has left the area for species which produce dormant liver parasites (especially P. vivax and P. ovale).
One of the main symptoms the patient will experience with a systemic infection is a fever. Caused by cytokine release, the fevers come in bursts (cycles) depending on when there is erythrocyte bursting (schizont rupture). Of course, this is different with the different species of plasmodium. P. knowlesi fevers occur in 24-hour waves, whereas P. vivax and P. ovale fevers occur in 48-hour waves and P. malariae fevers occur in 72-hour waves. P. falciparum is the odd one out with a more random fever cycle pattern. These fevers are called paroxysm fevers - fevers that occur in short bursts.
With the destruction of erythrocytes, an individual will present with the symptoms of haemolytic anaemia. These include:
- Pale in colour (pallor)
- Jaundice (release of haemoglobin from erythrocytes and thus build-up of bilirubin, its breakdown product)
- Dark coloured urine (also caused by presence of haemoglobin in urine)
- Weakness
- Dizziness and confusion
These features can occur in all Plasmodium infections, including P. falciparum, and are often more severe in falciparum malaria. Along with these symptoms of an uncomplicated malarial infection, P. falciparum creates a sticky protein that coats the surface of erythrocytes and causes them to clump together (like a clot) and occlude blood vessels leading to organ ischaemia. A variety of different organs are affected, and this would be a complicated malarial infection.
When malaria is suspected, diagnosis is confirmed by thick and thin blood films (x3) to test for the presence of parasites and then specifically which parasite is causing the infection (3 films are ordered as it is easy to produce false negative films depending on the sample and its timing). A full blood count is also ordered which should show a normocytic and normochromic anaemia. If it also shows a low platelet count (thrombocytopenia) and an increase in lactate dehydrogenase (from haemolysis) then the physician is likely to diagnose malaria.
Treatment
A complicated case of malaria will need immediate parenteral antimalarial therapy. This is usually a drug called artesunate and is given via IV (intravenously) or via IM (intramuscularly) injection.
This is usually given alongside quinine & doxycycline (P. falciparum) or chloroquine (P. vivax, ovale and malariae) - these drugs are also used to treat uncomplicated cases of malaria infection. Chloroquine is not used for P. falciparum due to widespread resistance.
If a physician thinks a patient has malaria but cannot confirm the species of Plasmodium, or even if the patient has malaria, then treat for chloroquine-resistant P. falciparum immediately.
Please see https://bnf.nice.org.uk/treatment-summary/malaria-treatment.html for up to date guidelines.
Let’s say the treatment plan didn’t work out too well and, although the patient was cured of malaria, they’ve got it again. This terminology for this recurrent malaria is split into three categories:
- Recrudescence (the treatment didn’t kill off the parasite)
- Relapse (patient is cleared of merozoites, but those dormant hypnozoites in the liver have decided to no longer be dormant)
- Reinfection (the patient was properly cured of malaria, but now has a completely new case of the same disease)
As with any disease, prevention is better than cure. When it comes to malaria, the answer lies in not letting mosquitoes bite people. This can be done by wearing clothing that covers your whole body, sleeping under mosquito nets at night and using insecticide sprays indoors. The Anopheles mosquito breeds in stagnant water, so community level control focuses on reducing breeding sites; for individuals, prevention relies on avoiding bites and using appropriate chemoprophylaxis.

Figure: Key things tourists should avoid and be advised to not do if travelling to an area with a high malaria risk
Public Domain Image National Library of Medicine - History of Medicine [No restrictions]
Legionellosis (Legionnaire’s Disease)
Legionellosis is a collective of conditions caused by Legionella bacteria, (Gram negative). The most serious condition Legionella can cause is known as Legionnaire’s disease.
Although Legionella bacteria are found right across rivers and lakes, the most likely place of infection is from somewhere where the water is at a high enough temperature to help the bacteria grow (somewhere like a poorly maintained shower system or air conditioning unit). This makes it likely to be caught in a poorly cleaned hotel or office rather than a home (which is very rare).
The bacteria are contracted by inhaling droplets of water that contain the bacteria. Once inside the body, L. pneumophilia invades macrophages in the lungs and replicates inside them.
Legionnaires' disease usually develops two to 10 days after exposure to legionella bacteria. It frequently begins with the following signs and symptoms:
- Headache
- Muscle/ chest pain
- Chills
- Fever that may be 40 oC or higher
And it can progress to include:
- Cough, which may bring up mucus and sometimes blood
- Shortness of breath
- Gastrointestinal symptoms, such as nausea, vomiting and diarrhoea
- Confusion or other mental changes
Legionnaires’ disease is an uncommon cause of pneumonia, but is readily treated with antibiotics (commonly, clarithromycin) if it is identified. Antibiotic treatment usually lasts one to three weeks and most people make a full recovery (but it might take a few weeks more to get back to normal).
Schistosomiasis is a parasitic disease caused by Schistosoma species, a group of flatworms common in tropical areas. Humans acquire the flatworms through unprotected skin contact in areas of fresh water contaminated with the helminths which have been released from snails (hence it’s other name of ‘snail fever’.
The symptoms patients present with are: similar to a fever, alongside:
- Diarrhoea
- Coughs
- Rashes
- Abdominal pain
- Muscle and joint pain
These symptoms are referred to as acute schistosomiasis.
Schistosomiasis also often involves chronic inflammation, causing damage due to the host’s immune response. Conditions caused include anaemia, cystitis (bladder irritation), cardiovascular and respiratory problems or neurological symptoms such as seizures and dizziness.
Diagnosis is confirmed by finding eggs in a urine or stool sample or by a blood test for antibodies.
Acute schistosomiasis symptoms are treated with a course of steroids to reduce inflammation. The curative treatment, praziquantel, is normally most effective once the worms have matured, so a patient might find that they aren’t given treatment until a number of weeks post-infection.
Also called enteric fever, typhoid fever is caused by Salmonella typhi (rod-shaped Gram negative bacteria). The usual presentation would be a picture of fever, diffuse abdominal pain and constipation. If left untreated, it may also cause delirium, GI haemorrhage and bowel perforation - even potential death a month post-infection!
Typhoid fever is most common where there is poor sanitation and limited access to clean water. Children are thought to be more likely to contract typhoid fever because their immune system is still developing, but the symptoms they present with tend to be less severe than with adults.
If the infection is diagnosed early and is mild, it can usually be treated at home with a seven to fourteen day course of oral antibiotics such as azithromycin. More severe disease is treated in hospital, often with intravenous ceftriaxone. The more serious infections will need to be treated by injections of antibiotics in hospital. Most patients will start to feel better after a few days.
There are two vaccinations against typhoid fever (a single injection or a course of three tablets) but neither offer 100% protection so it is important to take other precautions when travelling to certain areas (e.g. only consuming food and water you know is “clean”). Any cases in the UK are normally from patients that recently travelled in Bangladesh, India or Pakistan (although they aren’t the only places, with Asia, Africa and South America also being on the list of places a patient is likely to contract typhoid fever from).
Dengue fever or "breakbone fever", is caused by the dengue virus. The virus has four strains which are usually transmitted by mosquitoes (Aedes aegypti and Aedes albopictus).
The good news for people with dengue is that most infections only present with mild or even asymptomatic infections.
Around four to ten days after the bite, someone may suddenly present with:
- An intense headache
- Fever
- Muscle/joint pain (hence the name breakbone fever)
- A widespread red rash
Dengue normally resolves itself within a few days (which is good because there is no specific treatment for it other than to treat the symptoms as they present). Supportive treatments include taking paracetamol to relieve pain and fever and drinking plenty of fluids to avoid dehydration; non-steroidal anti-inflammatory drugs such as ibuprofen or aspirin should be avoided because of bleeding risk.
The bad news is that in 1-2% of infections (especially those in patients who have been previously infected by dengue), there is progression to severe dengue or dengue haemorrhagic fever which can be fatal. As with uncomplicated cases of dengue, the severe form has no cure so may require intensive care with IV fluids, oxygen therapy if required along with blood and platelet transfusions.
A patient can rapidly progress from standard dengue fever to dengue haemorrhagic fever, which can result in symptoms such as:
- Restlessness
- Acute, or sudden, fever
- Severe abdominal pain
- Bleeding or bruising under the skin (petechiae)
- Nosebleeds
- Large decreases in blood pressure (shock)
Along with bleeding from various places, this stage of dengue fever is characterised by low levels of platelets in the blood.
Attempts to increase platelet levels have led to claims that papaya can ‘cure’ dengue fever; however, there is no good quality evidence that papaya leaf extract is effective or safe for this purpose, and it is not recommended in clinical guidelines. Management remains supportive, with careful monitoring and treatment of complications. On the same line as this, if a patient was to be taking painkillers to help with the symptoms, then aspirin will only make matters worse (paracetamol is preferred). Aspirin and other NSAIDs increase the risk of bleeding because they impair platelet function, which is particularly dangerous in dengue.
Edited by: Dr. Ben Appleby
- 9243

