Next Lesson - Chronic Inflammation
Contents
- Clinical Signs of Inflammation
- Causes of Acute Inflammation
- Stages of Acute Inflammation
- Vascular Phase of Acute Inflammation
- Exudate
- Exudate Formation
- Types of Exudate
- The Role of Neutrophils in Acute Inflammation
- Phagocytosis
- A Summary of Chemical Mediators of Inflammation
- Local Complications of Acute Inflammation
- Systemic Effects of Acute Inflammation
- What Can Happen After Acute Inflammation?
- Quiz
Abstract
- There are five cardinal signs of inflammation: rubor, calor, tumor, dolor and functio laesa/ loss of function
- Acute inflammation can be broken down into:
- Stimulus e.g. infection, trauma or chemical agents.
- Vascular stage - slowing the local circulation and forming exudate at the site of inflammation.
- Cellular stage - the migration of neutrophils to the site.
- Either resolution where the tissue returns to normal, or persistence which can lead to chronic inflammation.
Core
Inflammation is the rapid response to injury of any vascularised living tissue in order to deliver various defensive materials such as white blood cells and proteins to the site of injury. The purpose of inflammation is to defend against infection and to clear damaged tissue.
The suffix '-itis' is used to indicate inflammation of an organ or tissue. For example, appendicitis is an inflammation of the appendix.
Acute inflammation is a type of inflammation which develops over the course of minutes, hours or days. If the inflammation lasts for weeks, months, or years and is associated with fibrotic changes then it is chronic inflammation.
Clinical Signs of Inflammation
There are 5 clinical signs of inflammation, which are as follows:
- Rubor - Redness
- Calor - Heat
- Tumor - Swelling
- Dolor - Pain
- Functio laesa - Loss of function
- This forces the person injured to immobilise the affected area which helps to reduce further damage.
This process can be remembered through the memory aid - Raging Cows Trampled Down Fred, or Really Can't Tolerate Dead Languages.
Common causes of acute inflammation include:
- Microbial infections e.g. pyogenic (pus causing) organisms
- Hypersensitivity reactions (in the acute phase)
- Foreign bodies e.g. splinters, sutures, dirt
- Tissue necrosis
- Physical trauma
- Chemical agents or radiation
Acute inflammation is innate (present from birth) and stereotyped (happens the same way no matter the cause of the inflammation), and is controlled by chemical mediators.
There are four stages of acute inflammation:
- Stimulus
- Vascular stage - slowing the circulation and forming exudate at the site of inflammation
- Cellular stage - the migration of neutrophils to the site
- Resolution or persistence
Vascular Phase of Acute Inflammation
The first phase of acute inflammation is the vascular phase, which occurs in the following steps:
- Vasoconstriction of arterioles (for a few seconds).
- Vasodilation of arterioles and then capillaries. This increases the blood flow to the affected area, resulting in heat and redness (calor and rubor).
- Increased permeability of local blood vessels allows proteins, cells and fluid to leave the blood vessel and enter the interstitial fluid causing swelling (tumor). This is called exudate
- The increased concentration of red blood cells in small vessels and increased blood viscosity leads to stasis within the vessel.
This process is controlled by different chemical mediators. Within the first minutes of inflammation, mediators such as histamine are released from mast cells, basophils and platelets. Histamine acts to dilate vasculature, increase vascular permeability and stimulate pain (dolor). As the inflammatory response continues it is mediated by chemicals known as leukotrienes and bradykinins.
Exudate is a collection of fluid, protein and cells that have left the blood vessel and formed in the interstitium. This exudate has several functions:
- To deliver fibrin, inflammatory mediators and immunoglobulins to the site of damage.
- Dilutes toxins to reduce the damage to tissues.
- Increases lymphatic drainage to deliver antigens and pathogens to lymph nodes to initiate an immune reaction.
The fluid flow across the vessel walls is controlled by a combination of hydrostatic pressure (the pressure exerted by a fluid which forces fluid out) and oncotic pressure (osmotic pressure exerted by proteins which draws fluid in). These are known as Starling’s Forces. This is a simplified description used for learning; more recent models place greater emphasis on endothelial permeability and the glycocalyx.
- If there is increased hydrostatic pressure, there is an increased flow of fluid out of the vessel.
- If there is increased oncotic pressure within the vessel then there will be a reduction in the flow of fluid out of the vessel.
- If there is an increased oncotic pressure in the interstitial fluid, there will be an increased flow of fluid out of the vessel as the oncotic pressure draws fluid out of the capillary.
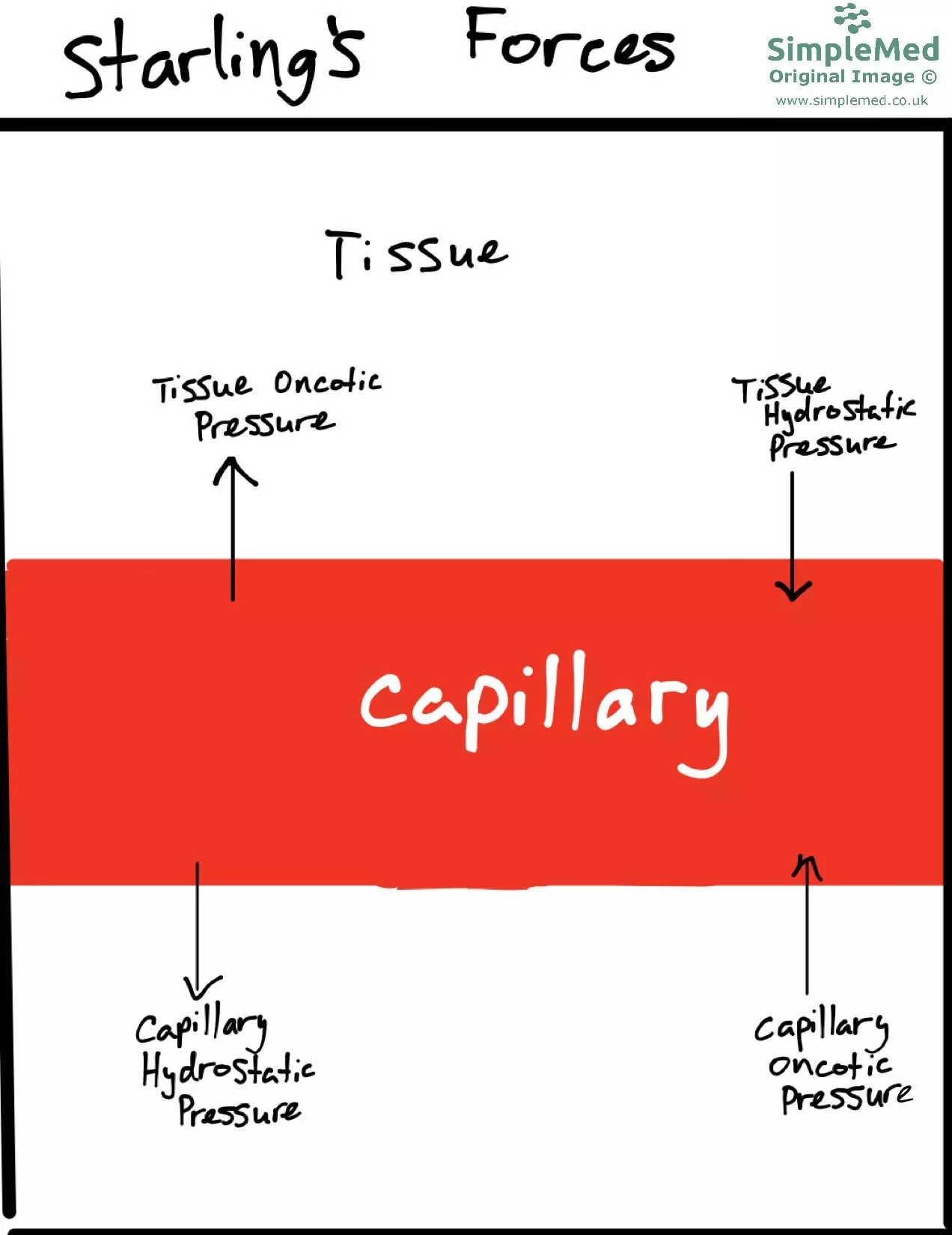
Diagram - The Starling forces and how they control the fluid movement into and out of the blood vessels
SimpleMed original by Benjamin Norris
Before exudate can form, the vasculature must become ‘leakier’ to allow proteins and fluid to cross more easily into the tissue space. Vascular leakage can occur due to the following mechanisms:
- Endothelial contraction forming gaps between cells.
- Mediated by histamine and leukotrienes.
- Cytoskeletal reorganisation forming gaps between cells.
- Mediated by cytokines, interleukin-1, and TNF (tumour necrosis factor).
- Direct injury - e.g. toxic burns, chemicals.
- Leucocyte dependent injury - damages endothelial cells causing gaps to form between them.
- Reactive oxygen species and enzymes from leucocytes.
- Increased transcytosis - production of channels within the endothelial cell to allow movement of proteins and fluid between cells.
- Mediated by VEGF.
Exudate forms via the following sequence of events:
- Arteriolar dilation (but unchanged venous tone) increases hydrostatic pressure in the capillaries.
- Increased vessel permeability by the above mechanisms causes movement of protein into the interstitium. This increases the oncotic pressure in the tissue and decreases the oncotic pressure in the vessels.
- The changes in oncotic and hydrostatic pressures result in a net flow of fluid out of the vessels, leading to tissue oedema.
- Pus/Abscess
- Creamy/white as it is rich in neutrophils. This is typical of infections of pyogenic bacteria.
- Haemorrhagic
- Red due to the presence of red blood cells. This indicates significant vascular damage.
- Serous
- Clear fluid with few leucocytes, suggesting no microorganisms. This exudate is produced in blisters and burns.
- Fibrinous
- Significant deposition of fibrin. This can result in friction between serosal surfaces which can be heard as a rubbing sound on auscultation e.g. in pericarditis.
The Role of Neutrophils in Acute Inflammation
The primary type of white blood cell involved in inflammation is the neutrophil, which is a type of granulocyte. Neutrophils are normally found in the blood and bone marrow, and their accumulation in tissues usually indicates the presence of a pathogenic organism or injury. They have a lifespan of 12-20 hours and cannot multiply.
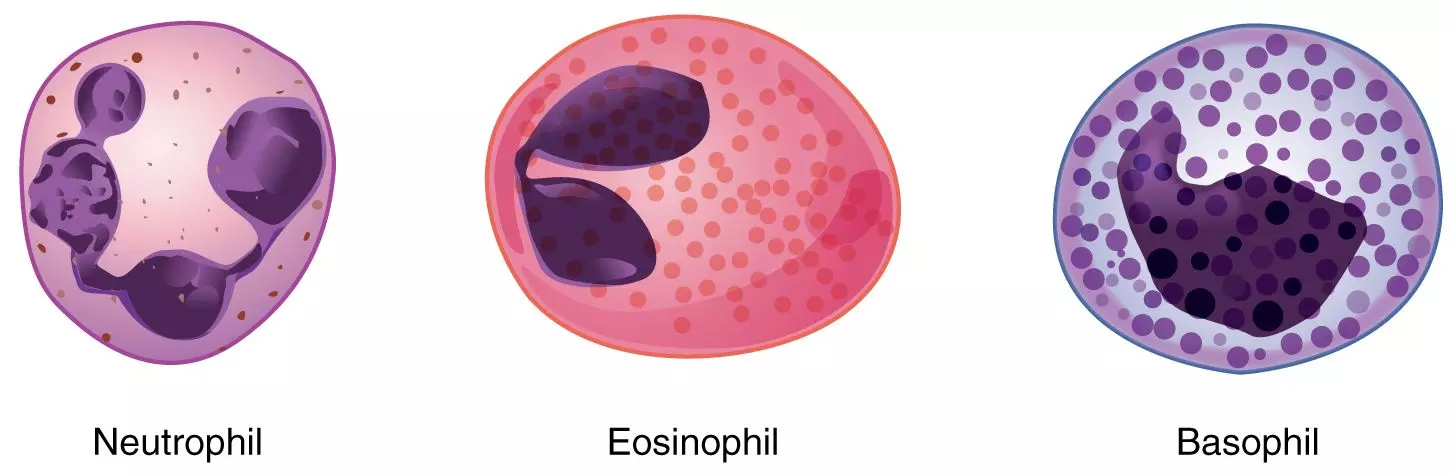
Diagram - The different granulocytes found in the body
Creative commons source by OpenStax College [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)]
To capture and kill a bacterium in the tissue space, a neutrophil must infiltrate the tissues. Neutrophils infiltrate the tissue and attach to bacteria through a series of overlapping steps:
- Chemotaxis - neutrophils are attracted towards the site of injury by chemical attractants.
- Activation - neutrophils switch to a higher metabolic level and change shape to help them move towards the chemical attractant.
- Margination - neutrophils move towards the endothelial wall where they then roll along it until they become trapped. When they become trapped, they then crawl out of the vessel.
- Diapedesis - neutrophils relax the junctions between the endothelial cells so they can move across the endothelium. They also use collagenase to break down the basement membrane.
- Recognition and attachment - neutrophils recognise the bacterium via the opsonins which attach to the bacterium. Neutrophils then move towards and attach to the bacterium.
- Opsonins are substances which make it easier for phagocytes to recognise targets. Examples include complement compounds and antibodies.
- Phagocytosis - this is the process by which the neutrophils engulf and destroy the bacteria.
Chemotaxis is the directional movement towards a chemical attractant. Both exogenous and endogenous substances can be chemotactic for leucocytes (see summary table). Within five seconds of the chemotaxin binding to the cell surface receptors the leucocyte undergoes conformational change to ‘activate’ it, which makes the cell more able to stick to microorganisms.
In most forms of acute inflammation neutrophils predominate in the inflammatory infiltrate during the first 6-24 hours. Neutrophils are replaced by monocytes in 24-48 hours.
Phagocytosis (cell eating) is the engulfment of solid particulate material by phagocytes, such as neutrophils. The process of phagocytosis is summarised in the diagram below.
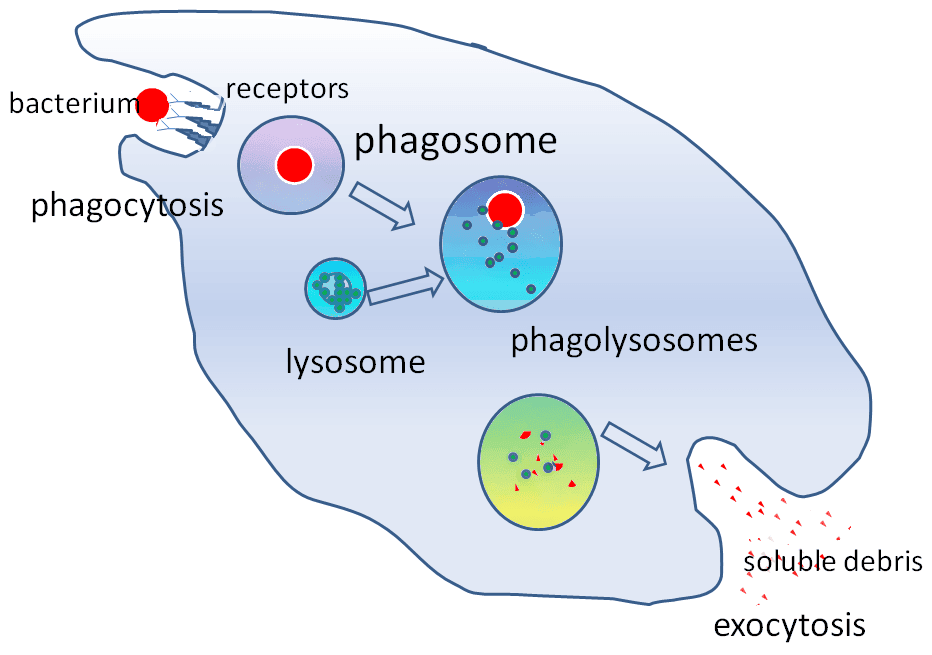
Diagram - The stages in phagocytosis
Creative commons source by Graham Colm [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)]
The membrane of the phagocyte forms a crater around the particle to be eaten. The edges then come together and the opposed plasma membranes fuse. The particle is then in a vacuole known as a phagosome. Lysosomes fuse with the phagosomes forming a phagolysosome. Chemicals are released that break down the engulfed particle. The debris is released by exocytosis.
Engulfed cells can be killed in one of two ways:
- O2 dependent mechanisms - release of oxygen-derived free radicals into the phagosome. The mechanism of killing is known as the oxidative burst.
- O2 independent mechanisms - uses enzymes to kill bacteria inside the phagolysosome e.g. proteases, lipases, nucleases.
A Summary of Chemical Mediators of Inflammation
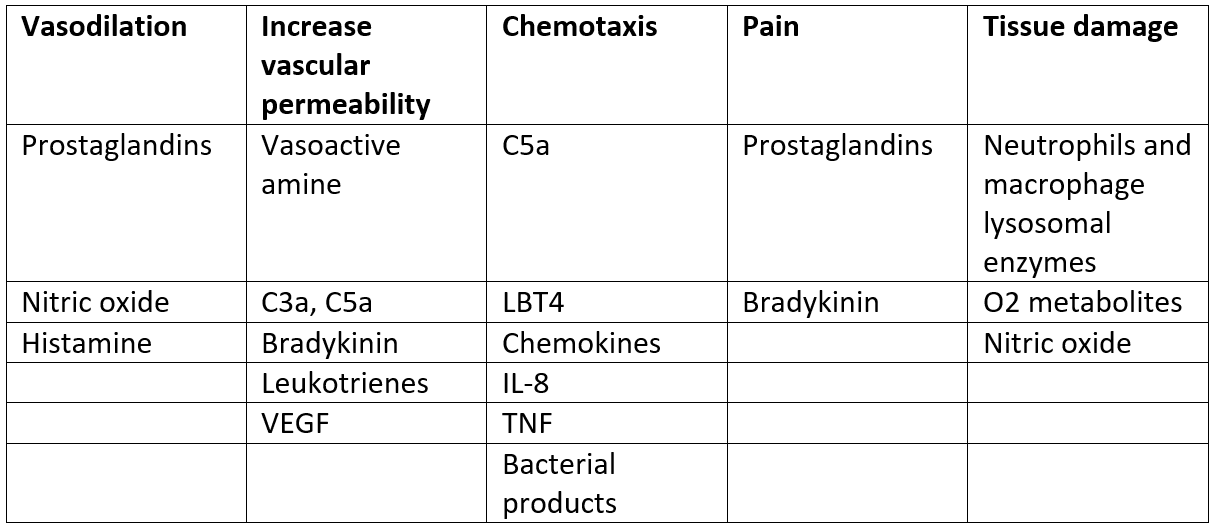
Table - The different chemical mediators in the body and the functions they have during inflammation
SimpleMed original by Benjamin Norris
Local Complications of Acute Inflammation
- Damage to normal tissue secondary to substances produced by neutrophils and released in phagocytosis
- Obstruction of tubes, e.g. bile duct, intestine due to compression by surrounding swelling
- Compression of vital structures secondary to swelling
- Loss of fluid as exudate, e.g. burns
- Pain and loss of function
Systemic Effects of Acute Inflammation
- Fever
- Pyrogenic substances are released that stimulate the hypothalamus to increase the body temperature e.g. IL-1 and TNF.
- Prostaglandins can also cause fever. Prostaglandin synthesis is inhibited by aspirin so aspirin can be used to help reduce fevers in adults.
- Leucocytosis
- IL-1 and TNF produce accelerated release of leucocytes from the bone marrow.
- Macrophages and T lymphocytes also stimulate the further release of other leucocytes.
- Acute phase response
- There is a change in the level of some plasma proteins as the liver changes its pattern of protein synthesis
- The response causes decreased appetite, increased pulse rate, altered sleep patterns and changes in plasma concentration of acute phase proteins such as:
- C-reactive protein (CRP) (clinically useful in assessing the level of inflammation)
- Alpha-1 antitrypsin
- Haptoglobin
- Fibrinogen
- Serum amyloid A protein
- Haemodynamic shock
- A clinical syndrome of circulatory failure, which can often be fatal.
- Can happen if bacterial products or inflammatory mediators spread around the body in the bloodstream, leading to body-wide inflammation.
- In shock there is a dramatic drop in blood pressure caused by widespread vasodilation and increased vascular permeability in response to the bacterial products in the blood.
What Can Happen After Acute Inflammation?
There are several possibilities following acute inflammation, including persisting and becoming chronic inflammation with fibrous repair. The acute inflammation could continue along with chronic inflammation (e.g. an abscess). The best scenario for acute inflammation is resolution.
With resolution the changes gradually reverse and vascular changes stop:
- Neutrophils no longer marginate
- Vessel permeability returns to normal
- Exudate drains to lymphatics
- Fibrin is degraded by plasmin and other proteases
- Neutrophils die, break up and are carried away or are engulfed
- Damaged tissue may be able to regenerate
Edited by: Dr. Bethany Turner
Reviewed by: Dr. Thomas Burnell
- 24261

