Next Lesson - Travel-related Infections
Abstract
- The term primary immunodeficiency disease is an umbrella term that encompasses more than 300 different clinical phenotypes.
- Primary immunodeficiency is a congenital disease caused by an intrinsic gene defect that alters the ability of the immune system to function correctly.
- Management of primary immunodeficiencies consists of supportive and specific treatments as well as treatment of any co-morbidities.
- Secondary immunodeficiency is an acquired condition as a result of an underlying disease or treatment.
Core
An immunocompromised host is a patient whose immune system is unable to respond appropriately and effectively to infectious microorganisms due to a defect in one or more components of the immune system.
The defect in the immune system can be congenital (i.e. due to an intrinsic gene defect) known as primary immunodeficiency, or acquired (i.e. as a result of an underlying disease or treatment) known as secondary immunodeficiency.
Due to the nature of the disease, and the vast array of different defects (>350 different mutations) that can lead to the condition, primary immunodeficiency is often poorly recognised and diagnosed. It is believed that in many cases, diagnosis is delayed for several years after the first onset of symptoms, and a significant proportion of patients are diagnosed in adulthood. This delay in diagnosis can leave patients with permanent tissue and organ damage by the time of diagnosis, hence the importance of awareness among clinicians to try and identify primary immunodeficiencies early to prevent damage and lead to a better prognosis and quality of life for the patient.
When should we suspect an Immunodeficiency?
The following mnemonic is often used as an initial guide when suspecting an immunodeficiency: SPUR
S- Severe: can the presenting complaint/recurring infection be life-threatening if untreated?
P- Persistent: does not respond appropriately to conventional treatment?
U- Unusual: is the site of infection and type of microbe unusual? E.g. deep tissue infection, opportunistic microbe
R- Recurrent: does the patient have more frequent infections than the average for a person without any immunocompromised features?
If the history and features of infections a patient presents with fulfil the above criteria, healthcare professionals should suspect immunodeficiency.
Alternatively, the Jeffrey Modell Foundation Medical Advisory Board developed a series of 10 warning signs to recognise PID (Primary Immunodeficiency):

Figure: The 10 warning signs for PID in adults and children
SimpleMed original by Dr. Elena Perez
This model has a few limitations: despite being developed by a series of experts in the field of PIDs, there is still a lack of population-based evidence to support it. It is also worth noting that some PID patients might have non-infectious manifestations such as autoimmunity, malignancy or inflammatory responses to be aware of. Different variations of PID present with different rates of given malignancies.
If immunodeficiency is suspected, the next step is to carry out some laboratory investigations to confirm the type of immunodeficiency:
- It is important to review the past medical history for any potential cause of secondary immunodeficiency
- Carry out a full blood count (FBC)
- Test humoral (antibody) immunity
- IgG, IgA, IgM (+/- IgE)
- IgG1-4 subclasses
- IgG levels to specific previous vaccines e.g. tetanus toxoid, measles, mumps, rubella
- Measure antibody in response to ‘test’ immunisation
- Test for cell-mediated immunity
- Lymphocyte count
- Lymphocyte subset analysis (CD4+, CD8+, T, NK, and B cells) extended marker panel
- In vitro tests of T cell function
- Test for phagocytic cells
- Neutrophil count
- Neutrophil function tests e.g. oxidative burst for CGD (Chronic Granulomatous Disease)
- Adhesion molecule expression (for LAD: Leukocyte Adhesion Deficiency)
- Test for complement
- Individual components
- Test of complement function (CH50/AP50: classical and alternative complement pathways)
- Definitive tests - molecular testing and gene mutations.
As mentioned above, the term primary immunodeficiency refers to congenital immunodeficiencies that are due to intrinsic gene defects. These can lead to a missing protein, cell or non-functional components of the immune system, making it weak or dysfunctional.
The main immunodeficiencies that we will focus on are:
- Predominantly antibody deficiencies - account for about 65% of all PIDs.
- Common variable immunodeficiency (CVID)- the patient has B cells but they produce reduced or dysfunctional antibodies
- Bruton’s disease (X-linked agammaglobulinaemia, XLA)- the patients lack B cells so can not produce antibodies
- IgA deficiency- the most common primary immunodeficiency. Around 90% of individuals are asymptomatic and prevalence is between 1 in 100 to 1 in 1000 in the general population. These patients have an increased risk of autoimmune conditions.
- Hyper IgM- failure of immunoglobulin class switching in B cells, resulting in low IgG, IgA and IgE levels with normal or raised IgM.
- Combined T and B cell deficiencies - represent around 15% of all PIDs and result in severe combined immunodeficiency (SCID)- as T cells are defective B cells are not activated
- Phagocytic defects - responsible for about 10% of all PIDs which lead to chronic granulomatous disease (CGD) - phagocytes lack killing machinery and are ineffective as a result
- Other cellular immunodeficiencies - make up 5-10% of all PIDs, the most common being DiGeorge syndrome (a deletion syndrome on chromosome 22q11.2) with an absent or hypoplastic thymus and no T cell maturation.
A way of differentiating between the different types of primary immunodeficiencies is by looking at the age of onset of first symptoms:
- Onset < 6 months old: most likely a T-cell or phagocyte defect.
- Onset > 6 months old < 5 years old: suggests a B-cell/antibody or phagocyte defect (as now maternal antibodies have been cleared so antibody defects will become clear).
- Onset > 5 years old: most likely a B-cell/antibody/complement or a secondary immunodeficiency.
Another way of differentiating between the major immune defects is by looking at the presentation of the infections (type of microbe and site) as specific presentations are associated with specific defects of components of the immune system:
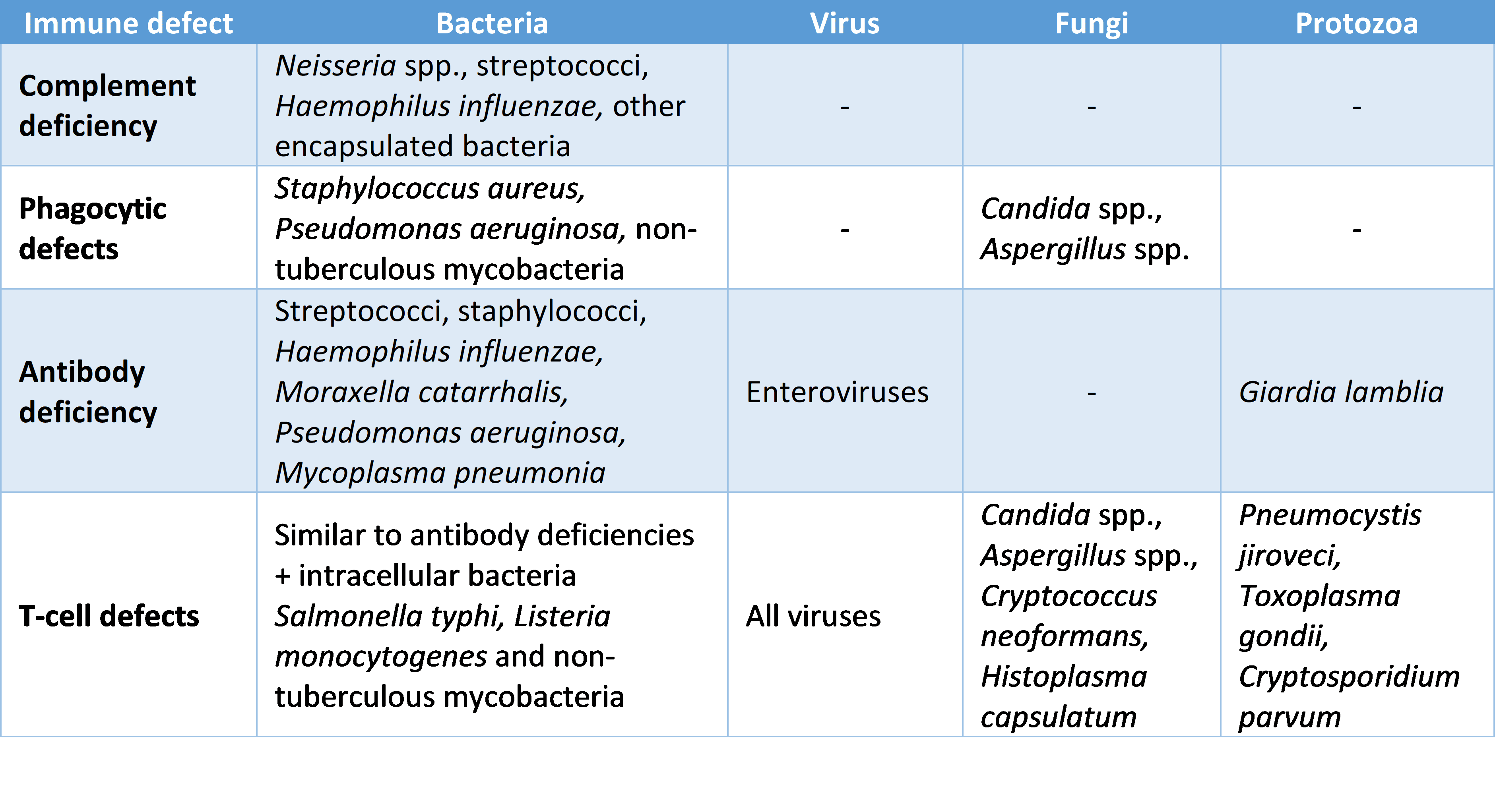
Figure: Infections associated with the different types of immune system defects
SimpleMed original by Dr. Elena Perez
Management of primary immunodeficiency diseases has three components: supportive treatment, specific treatment and management of any co-morbidities.
- Supportive treatment:
- Infection prevention: prophylactic antibiotics
- Treat infections promptly and aggressively, alongside immunoglobulin replacement where indicated
- Nutritional support: vitamins A/D
- Use UV-irradiated CMV -ve blood products only
- Avoid live attenuated vaccines in patients with severe PIDs (e.g. SCID)
- Specific treatment:
- Regular immunoglobulin therapy: IVIg or ScIg (young patients)
- SCID: haematopoietic stem cell therapy (HSCT, 90% success)
- Treat comorbidities:
- Autoimmune and malignancy treatment if disease arises
- Assess organ damage (lung function assessment)
- Avoid non-essential exposure to radiation
Secondary immune deficiencies are the result of a decreased production of immune components. The single most common cause of secondary immunodeficiency worldwide is malnutrition. Other reasons might be as a result of infections (see article on HIV), liver diseases, lymphoproliferative diseases or asplenism.
The spleen is an important organ of the immune system and by understanding its immune functions, one can easily understand how its removal or damage would contribute to secondary immunodeficiency. The spleen has an important role in:
- Removal of blood-borne pathogens: especially in dealing with encapsulated bacteria.
- Antibody production: during the acute phase response it produces IgM, and for long-term protection it produces IgG.
- Splenic macrophages: removal of opsonized microbes, removal of immune complexes.
Asplenic or splenectomised patients thus need to be especially careful regarding infections and hospital admissions, so they are often given life-long penicillin prophylaxis and immunisation against encapsulated bacteria (Neisseria meningitidis, Haemophilus influenza and Streptococcus pneumonia). In addition, they often wear a Medic Alert bracelet to inform healthcare professionals in emergencies.
Similarly, patients with haematological malignancies or who have undergone chemotherapy are highly susceptible to infections. This is due to chemotherapy-induced neutropenia and damage to mucosal barriers. Commonly used vascular catheters breach the skin and provide a route to let bacteria into the body as well as the attraction for biofilms which can also increase the risk of infection. Any suspected neutropenic sepsis should be treated as a medical emergency and the patient should be given empiric antibiotic therapy immediately.
Finally, any condition that results in an increased loss or catabolism of immune components can also cause secondary immunodeficiency. Some examples of such conditions include protein-losing diseases such as nephropathy or enteropathy and burns.
Examples of common manifestations of secondary immunodeficiency:
Shingles, or Varicella Zoster Virus (VZV) infection, is an example of a common infection in secondary immunodeficiency. Primary infection with Varicella zoster (chickenpox) is usually disseminated and remains latent by establishing itself in multiple sensory dorsal root ganglia. Reactivation of latent Herpes zoster commonly occurs in immunocompromised patients and manifests itself as shingles, which appears as a painful rash of papules (blisters) with erythema (redness) that usually follows dermatomal patterns. It is treated with pain relief and antivirals such as aciclovir.
Candida albicans is known as an opportunistic pathogen: it can be commonly found in the normal flora of the mouth and gut of healthy hosts. However, in the immunocompromised, it can become pathogenic and overgrowth of the yeast can cause candidiasis. Oral candidiasis is indeed a characteristic sign of HIV development into AIDS.
Finally, Aspergillus, a genus of conidial fungi, that comprises human and crop pathogen species, is also an organism characteristically isolated from immunocompromised hosts. Aspergillus fumigatus is the most common organism causing aspergillosis. It primarily colonises paranasal sinuses and lungs, potentially leading to necrotising pneumonia. Diagnosis of pulmonary aspergillosis can be made from the characteristic ‘halo’ signs on HRCT scans.
Edited by: Dr. Ben Appleby and Dr. Thomas Burnell
Reviewed by: Dr. Marcus Judge
- 10508

